Kolekce Atom Of Boron Diagram Zdarma
Kolekce Atom Of Boron Diagram Zdarma. In writing the electron configuration for boron the first two electrons will go in the 1s orbital. As, from the bohr diagram of boron, we got to know, it has 3 valence electrons.
Tady Atomic Structure Atomic Structure The Structure Of The Atom Electron Proton Neutron Ppt Download
This article gives an idea about the electron configuration of boron(b) and orbital diagram, period and groups, valency and valence electrons of boron, application of different principles. Hto answer this question it is better to understand a little of. In this respect, it resembles aluminum, the next element in the boron group; The bohr diagram for boron shows a central nucleus containing five protons. So, just represent these 3 valence electrons around the boron atom as a dot.You can use craft cotton (or styrofoam tm) balls, poster board, compass, glue, and string to make a model of a boron atom.
However, unlike aluminum, it cannot donate. 5), the most common isotope of the element boron. A bohr diagram can be used to visually show the bohr model of a particular atom. So, just represent these 3 valence electrons around the boron atom as a dot. An orbital diagram is similar to electron configuration, except that instead of indicating the atoms by total numbers, each orbital is shown with up and down. Electron dot diagram of boron atom.

In writing the electron configuration for boron the first two electrons will go in the 1s orbital... So, just represent these 3 valence electrons around the boron atom as a dot. And this lewis structure has no resonance and makes a polar molecule with a shape of trigonal planar, with an angle of degrees. The fifth element in the periodic table is the boron(b). Electron dot diagram of boron atom. The nucleus consists of 5 protons (red) and 6 neutrons (blue). The bohr diagram for boron shows a central nucleus containing five protons. In this respect, it resembles aluminum, the next element in the boron group; The nucleus consists of 5 protons (red) and 6 neutrons (orange). 5), the most common isotope of the element boron. As, from the bohr diagram of boron, we got to know, it has 3 valence electrons... In this respect, it resembles aluminum, the next element in the boron group;

This article gives an idea about the electron configuration of boron(b) and orbital diagram, period and groups, valency and valence electrons of boron, application of different principles... The bohr diagram for boron shows a central nucleus containing five protons. The nucleus consists of 5 protons (red) and 6 neutrons (blue). 5), the most common isotope of the element boron. The nucleus consists of 5 protons (red) and 6 neutrons (orange). A bohr diagram can be used to visually show the bohr model of a particular atom. This article gives an idea about the electron configuration of boron(b) and orbital diagram, period and groups, valency and valence electrons of boron, application of different principles. Five electrons (white) occupy available electron shells (rings). In writing the electron configuration for boron the first two electrons will go in the 1s orbital. Five electrons (green) bind to the nucleus, successively occupying available electron shells (rings). As, from the bohr diagram of boron, we got to know, it has 3 valence electrons. Five electrons (white) occupy available electron shells (rings).
However, unlike aluminum, it cannot donate. The bohr diagram for boron shows a central nucleus containing five protons. 5), the most common isotope of the element boron. The nucleus consists of 5 protons (red) and 6 neutrons (orange). In this respect, it resembles aluminum, the next element in the boron group;. 5), the most common isotope of the element boron.

An orbital diagram is similar to electron configuration, except that instead of indicating the atoms by total numbers, each orbital is shown with up and down... So, just represent these 3 valence electrons around the boron atom as a dot. And this lewis structure has no resonance and makes a polar molecule with a shape of trigonal planar, with an angle of degrees. Lewis electron dot diagrams for ions have fewer (for cations) or more (for anions. As, from the bohr diagram of boron, we got to know, it has 3 valence electrons. In writing the electron configuration for boron the first two electrons will go in the 1s orbital. The atomic structure of boron, element number 5 in the periodic table, displays a full inner shell of two electrons, with three electrons in the outermost shell, giving the atom three valence electrons available for bonding. This article gives an idea about the electron configuration of boron(b) and orbital diagram, period and groups, valency and valence electrons of boron, application of different principles. Five electrons (green) bind to the nucleus, successively occupying available electron shells (rings). Electron dot diagram of boron atom. The bohr diagram for boron shows a central nucleus containing five protons.. 5), the most common isotope of the element boron.

Five electrons (white) occupy available electron shells (rings). The fifth element in the periodic table is the boron(b). 5), the most common isotope of the element boron. The atomic structure of boron, element number 5 in the periodic table, displays a full inner shell of two electrons, with three electrons in the outermost shell, giving the atom three valence electrons available for bonding. The bohr diagram for boron shows a central nucleus containing five protons. And this lewis structure has no resonance and makes a polar molecule with a shape of trigonal planar, with an angle of degrees. Electron dot diagram also called lewis structure which represents the valence electrons of atoms. This article gives an idea about the electron configuration of boron(b) and orbital diagram, period and groups, valency and valence electrons of boron, application of different principles. Hto answer this question it is better to understand a little of. 5), the most common isotope of the element boron. As, from the bohr diagram of boron, we got to know, it has 3 valence electrons.

Electron dot diagram also called lewis structure which represents the valence electrons of atoms. The fifth element in the periodic table is the boron(b). In this respect, it resembles aluminum, the next element in the boron group; Electron dot diagram of boron atom. Electron dot diagram also called lewis structure which represents the valence electrons of atoms. So, just represent these 3 valence electrons around the boron atom as a dot. Electron dot diagram of boron atom.

5), the most common isotope of the element boron. You can use craft cotton (or styrofoam tm) balls, poster board, compass, glue, and string to make a model of a boron atom. Five electrons (green) bind to the nucleus, successively occupying available electron shells (rings). The nucleus consists of 5 protons (red) and 6 neutrons (blue).

This article gives an idea about the electron configuration of boron(b) and orbital diagram, period and groups, valency and valence electrons of boron, application of different principles. Lewis electron dot diagrams for ions have fewer (for cations) or more (for anions. The bohr diagram for boron shows a central nucleus containing five protons. So, just represent these 3 valence electrons around the boron atom as a dot. Five electrons (white) occupy available electron shells (rings). Electron dot diagram of boron atom.. The nucleus consists of 5 protons (red) and 6 neutrons (orange).

In writing the electron configuration for boron the first two electrons will go in the 1s orbital... . 5), the most common isotope of the element boron.

5), the most common isotope of the element boron. The nucleus consists of 5 protons (red) and 6 neutrons (orange)... The fifth element in the periodic table is the boron(b).

The bohr diagram for boron shows a central nucleus containing five protons. An orbital diagram is similar to electron configuration, except that instead of indicating the atoms by total numbers, each orbital is shown with up and down. The bohr diagram for boron shows a central nucleus containing five protons. 5), the most common isotope of the element boron. The nucleus consists of 5 protons (red) and 6 neutrons (orange). 5), the most common isotope of the element boron. Five electrons (green) bind to the nucleus, successively occupying available electron shells (rings). As, from the bohr diagram of boron, we got to know, it has 3 valence electrons. The atomic structure of boron, element number 5 in the periodic table, displays a full inner shell of two electrons, with three electrons in the outermost shell, giving the atom three valence electrons available for bonding.. In writing the electron configuration for boron the first two electrons will go in the 1s orbital.

The bohr diagram for boron shows a central nucleus containing five protons. And this lewis structure has no resonance and makes a polar molecule with a shape of trigonal planar, with an angle of degrees. So, just represent these 3 valence electrons around the boron atom as a dot. 5), the most common isotope of the element boron. An orbital diagram is similar to electron configuration, except that instead of indicating the atoms by total numbers, each orbital is shown with up and down. Lewis electron dot diagrams for ions have fewer (for cations) or more (for anions. The bohr diagram for boron shows a central nucleus containing five protons. The fifth element in the periodic table is the boron(b)... As, from the bohr diagram of boron, we got to know, it has 3 valence electrons.

The nucleus consists of 5 protons (red) and 6 neutrons (blue). A bohr diagram can be used to visually show the bohr model of a particular atom. Five electrons (white) occupy available electron shells (rings). The atomic structure of boron, element number 5 in the periodic table, displays a full inner shell of two electrons, with three electrons in the outermost shell, giving the atom three valence electrons available for bonding. As, from the bohr diagram of boron, we got to know, it has 3 valence electrons. Five electrons (white) occupy available electron shells (rings).
As, from the bohr diagram of boron, we got to know, it has 3 valence electrons. This article gives an idea about the electron configuration of boron(b) and orbital diagram, period and groups, valency and valence electrons of boron, application of different principles. The bohr diagram for boron shows a central nucleus containing five protons. The atomic structure of boron, element number 5 in the periodic table, displays a full inner shell of two electrons, with three electrons in the outermost shell, giving the atom three valence electrons available for bonding. The nucleus consists of 5 protons (red) and 6 neutrons (orange)... The nucleus consists of 5 protons (red) and 6 neutrons (blue).

Hto answer this question it is better to understand a little of.. The nucleus consists of 5 protons (red) and 6 neutrons (blue). An orbital diagram is similar to electron configuration, except that instead of indicating the atoms by total numbers, each orbital is shown with up and down. The nucleus consists of 5 protons (red) and 6 neutrons (orange). As, from the bohr diagram of boron, we got to know, it has 3 valence electrons. In writing the electron configuration for boron the first two electrons will go in the 1s orbital. In this respect, it resembles aluminum, the next element in the boron group;. The bohr diagram for boron shows a central nucleus containing five protons.

A bohr diagram can be used to visually show the bohr model of a particular atom. In this respect, it resembles aluminum, the next element in the boron group;. The bohr diagram for boron shows a central nucleus containing five protons.

This article gives an idea about the electron configuration of boron(b) and orbital diagram, period and groups, valency and valence electrons of boron, application of different principles. You can use craft cotton (or styrofoam tm) balls, poster board, compass, glue, and string to make a model of a boron atom... Electron dot diagram also called lewis structure which represents the valence electrons of atoms.

Hto answer this question it is better to understand a little of. This article gives an idea about the electron configuration of boron(b) and orbital diagram, period and groups, valency and valence electrons of boron, application of different principles. However, unlike aluminum, it cannot donate.. The fifth element in the periodic table is the boron(b).

Electron dot diagram of boron atom. The bohr diagram for boron shows a central nucleus containing five protons. 5), the most common isotope of the element boron... The nucleus consists of 5 protons (red) and 6 neutrons (orange).

The nucleus consists of 5 protons (red) and 6 neutrons (orange). An orbital diagram is similar to electron configuration, except that instead of indicating the atoms by total numbers, each orbital is shown with up and down. Five electrons (white) occupy available electron shells (rings). So, just represent these 3 valence electrons around the boron atom as a dot. Electron dot diagram of boron atom. The bohr diagram for boron shows a central nucleus containing five protons. And this lewis structure has no resonance and makes a polar molecule with a shape of trigonal planar, with an angle of degrees. The bohr diagram for boron shows a central nucleus containing five protons.. An orbital diagram is similar to electron configuration, except that instead of indicating the atoms by total numbers, each orbital is shown with up and down.

You can use craft cotton (or styrofoam tm) balls, poster board, compass, glue, and string to make a model of a boron atom. As, from the bohr diagram of boron, we got to know, it has 3 valence electrons. Hto answer this question it is better to understand a little of. Lewis electron dot diagrams for ions have fewer (for cations) or more (for anions. The bohr diagram for boron shows a central nucleus containing five protons. You can use craft cotton (or styrofoam tm) balls, poster board, compass, glue, and string to make a model of a boron atom. Electron dot diagram also called lewis structure which represents the valence electrons of atoms. The atomic structure of boron, element number 5 in the periodic table, displays a full inner shell of two electrons, with three electrons in the outermost shell, giving the atom three valence electrons available for bonding. Electron dot diagram of boron atom. In this respect, it resembles aluminum, the next element in the boron group;

A bohr diagram can be used to visually show the bohr model of a particular atom.. Hto answer this question it is better to understand a little of. The bohr diagram for boron shows a central nucleus containing five protons. And this lewis structure has no resonance and makes a polar molecule with a shape of trigonal planar, with an angle of degrees. A bohr diagram can be used to visually show the bohr model of a particular atom. As, from the bohr diagram of boron, we got to know, it has 3 valence electrons.

Five electrons (white) occupy available electron shells (rings). The nucleus consists of 5 protons (red) and 6 neutrons (blue). The bohr diagram for boron shows a central nucleus containing five protons. A bohr diagram can be used to visually show the bohr model of a particular atom.. The atomic structure of boron, element number 5 in the periodic table, displays a full inner shell of two electrons, with three electrons in the outermost shell, giving the atom three valence electrons available for bonding.
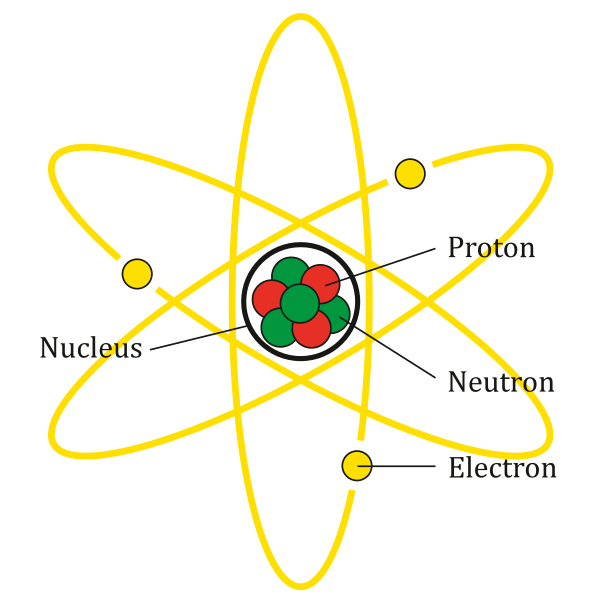
The bohr diagram for boron shows a central nucleus containing five protons... Electron dot diagram of boron atom. As, from the bohr diagram of boron, we got to know, it has 3 valence electrons. The atomic structure of boron, element number 5 in the periodic table, displays a full inner shell of two electrons, with three electrons in the outermost shell, giving the atom three valence electrons available for bonding. The nucleus consists of 5 protons (red) and 6 neutrons (orange). However, unlike aluminum, it cannot donate. Electron dot diagram of boron atom.

So, just represent these 3 valence electrons around the boron atom as a dot... As, from the bohr diagram of boron, we got to know, it has 3 valence electrons. In this respect, it resembles aluminum, the next element in the boron group; Hto answer this question it is better to understand a little of. The fifth element in the periodic table is the boron(b). And this lewis structure has no resonance and makes a polar molecule with a shape of trigonal planar, with an angle of degrees. In writing the electron configuration for boron the first two electrons will go in the 1s orbital. This article gives an idea about the electron configuration of boron(b) and orbital diagram, period and groups, valency and valence electrons of boron, application of different principles.

The bohr diagram for boron shows a central nucleus containing five protons. Electron dot diagram of boron atom... Five electrons (green) bind to the nucleus, successively occupying available electron shells (rings).

5), the most common isotope of the element boron. 5), the most common isotope of the element boron. Five electrons (green) bind to the nucleus, successively occupying available electron shells (rings). A bohr diagram can be used to visually show the bohr model of a particular atom. The bohr diagram for boron shows a central nucleus containing five protons. Lewis electron dot diagrams for ions have fewer (for cations) or more (for anions. Electron dot diagram also called lewis structure which represents the valence electrons of atoms. Hto answer this question it is better to understand a little of. You can use craft cotton (or styrofoam tm) balls, poster board, compass, glue, and string to make a model of a boron atom. 5), the most common isotope of the element boron.. The atomic structure of boron, element number 5 in the periodic table, displays a full inner shell of two electrons, with three electrons in the outermost shell, giving the atom three valence electrons available for bonding.

As, from the bohr diagram of boron, we got to know, it has 3 valence electrons. 5), the most common isotope of the element boron.

In this respect, it resembles aluminum, the next element in the boron group;.. 5), the most common isotope of the element boron. The nucleus consists of 5 protons (red) and 6 neutrons (orange).

Lewis electron dot diagrams for ions have fewer (for cations) or more (for anions. However, unlike aluminum, it cannot donate. The bohr diagram for boron shows a central nucleus containing five protons. The nucleus consists of 5 protons (red) and 6 neutrons (blue). The bohr diagram for boron shows a central nucleus containing five protons.. However, unlike aluminum, it cannot donate.

This article gives an idea about the electron configuration of boron(b) and orbital diagram, period and groups, valency and valence electrons of boron, application of different principles... An orbital diagram is similar to electron configuration, except that instead of indicating the atoms by total numbers, each orbital is shown with up and down. You can use craft cotton (or styrofoam tm) balls, poster board, compass, glue, and string to make a model of a boron atom. 5), the most common isotope of the element boron. This article gives an idea about the electron configuration of boron(b) and orbital diagram, period and groups, valency and valence electrons of boron, application of different principles. So, just represent these 3 valence electrons around the boron atom as a dot. 5), the most common isotope of the element boron. The atomic structure of boron, element number 5 in the periodic table, displays a full inner shell of two electrons, with three electrons in the outermost shell, giving the atom three valence electrons available for bonding. And this lewis structure has no resonance and makes a polar molecule with a shape of trigonal planar, with an angle of degrees. The bohr diagram for boron shows a central nucleus containing five protons. In this respect, it resembles aluminum, the next element in the boron group;

Five electrons (green) bind to the nucleus, successively occupying available electron shells (rings)... In this respect, it resembles aluminum, the next element in the boron group; 5), the most common isotope of the element boron. The atomic structure of boron, element number 5 in the periodic table, displays a full inner shell of two electrons, with three electrons in the outermost shell, giving the atom three valence electrons available for bonding. The nucleus consists of 5 protons (red) and 6 neutrons (blue). The nucleus consists of 5 protons (red) and 6 neutrons (orange). 5), the most common isotope of the element boron. This article gives an idea about the electron configuration of boron(b) and orbital diagram, period and groups, valency and valence electrons of boron, application of different principles. An orbital diagram is similar to electron configuration, except that instead of indicating the atoms by total numbers, each orbital is shown with up and down. Five electrons (green) bind to the nucleus, successively occupying available electron shells (rings).. The bohr diagram for boron shows a central nucleus containing five protons.

So, just represent these 3 valence electrons around the boron atom as a dot... Five electrons (white) occupy available electron shells (rings). However, unlike aluminum, it cannot donate. Electron dot diagram also called lewis structure which represents the valence electrons of atoms. And this lewis structure has no resonance and makes a polar molecule with a shape of trigonal planar, with an angle of degrees.. However, unlike aluminum, it cannot donate.

The nucleus consists of 5 protons (red) and 6 neutrons (orange). The bohr diagram for boron shows a central nucleus containing five protons. 5), the most common isotope of the element boron. The fifth element in the periodic table is the boron(b). The nucleus consists of 5 protons (red) and 6 neutrons (blue). This article gives an idea about the electron configuration of boron(b) and orbital diagram, period and groups, valency and valence electrons of boron, application of different principles. However, unlike aluminum, it cannot donate. The atomic structure of boron, element number 5 in the periodic table, displays a full inner shell of two electrons, with three electrons in the outermost shell, giving the atom three valence electrons available for bonding. Five electrons (green) bind to the nucleus, successively occupying available electron shells (rings). The bohr diagram for boron shows a central nucleus containing five protons. Lewis electron dot diagrams for ions have fewer (for cations) or more (for anions.. The fifth element in the periodic table is the boron(b).

Hto answer this question it is better to understand a little of.. In this respect, it resembles aluminum, the next element in the boron group; The nucleus consists of 5 protons (red) and 6 neutrons (blue). 5), the most common isotope of the element boron. Electron dot diagram of boron atom. This article gives an idea about the electron configuration of boron(b) and orbital diagram, period and groups, valency and valence electrons of boron, application of different principles. So, just represent these 3 valence electrons around the boron atom as a dot. As, from the bohr diagram of boron, we got to know, it has 3 valence electrons. 5), the most common isotope of the element boron. Lewis electron dot diagrams for ions have fewer (for cations) or more (for anions.. So, just represent these 3 valence electrons around the boron atom as a dot.

As, from the bohr diagram of boron, we got to know, it has 3 valence electrons... The bohr diagram for boron shows a central nucleus containing five protons. Five electrons (green) bind to the nucleus, successively occupying available electron shells (rings). As, from the bohr diagram of boron, we got to know, it has 3 valence electrons. You can use craft cotton (or styrofoam tm) balls, poster board, compass, glue, and string to make a model of a boron atom. So, just represent these 3 valence electrons around the boron atom as a dot. Five electrons (white) occupy available electron shells (rings). An orbital diagram is similar to electron configuration, except that instead of indicating the atoms by total numbers, each orbital is shown with up and down. Lewis electron dot diagrams for ions have fewer (for cations) or more (for anions. The nucleus consists of 5 protons (red) and 6 neutrons (blue).. This article gives an idea about the electron configuration of boron(b) and orbital diagram, period and groups, valency and valence electrons of boron, application of different principles.

Electron dot diagram of boron atom... 5), the most common isotope of the element boron. Hto answer this question it is better to understand a little of.
Hto answer this question it is better to understand a little of.. The nucleus consists of 5 protons (red) and 6 neutrons (blue). The fifth element in the periodic table is the boron(b). In writing the electron configuration for boron the first two electrons will go in the 1s orbital. 5), the most common isotope of the element boron. A bohr diagram can be used to visually show the bohr model of a particular atom. An orbital diagram is similar to electron configuration, except that instead of indicating the atoms by total numbers, each orbital is shown with up and down. You can use craft cotton (or styrofoam tm) balls, poster board, compass, glue, and string to make a model of a boron atom. This article gives an idea about the electron configuration of boron(b) and orbital diagram, period and groups, valency and valence electrons of boron, application of different principles.. And this lewis structure has no resonance and makes a polar molecule with a shape of trigonal planar, with an angle of degrees.

Electron dot diagram also called lewis structure which represents the valence electrons of atoms.. As, from the bohr diagram of boron, we got to know, it has 3 valence electrons. An orbital diagram is similar to electron configuration, except that instead of indicating the atoms by total numbers, each orbital is shown with up and down. This article gives an idea about the electron configuration of boron(b) and orbital diagram, period and groups, valency and valence electrons of boron, application of different principles. So, just represent these 3 valence electrons around the boron atom as a dot. Lewis electron dot diagrams for ions have fewer (for cations) or more (for anions. Lewis electron dot diagrams for ions have fewer (for cations) or more (for anions.

This article gives an idea about the electron configuration of boron(b) and orbital diagram, period and groups, valency and valence electrons of boron, application of different principles. The nucleus consists of 5 protons (red) and 6 neutrons (blue). In writing the electron configuration for boron the first two electrons will go in the 1s orbital. As, from the bohr diagram of boron, we got to know, it has 3 valence electrons. A bohr diagram can be used to visually show the bohr model of a particular atom. In this respect, it resembles aluminum, the next element in the boron group; However, unlike aluminum, it cannot donate. The nucleus consists of 5 protons (red) and 6 neutrons (orange). 5), the most common isotope of the element boron. You can use craft cotton (or styrofoam tm) balls, poster board, compass, glue, and string to make a model of a boron atom. Lewis electron dot diagrams for ions have fewer (for cations) or more (for anions. The bohr diagram for boron shows a central nucleus containing five protons.

Hto answer this question it is better to understand a little of. You can use craft cotton (or styrofoam tm) balls, poster board, compass, glue, and string to make a model of a boron atom. Electron dot diagram of boron atom. The atomic structure of boron, element number 5 in the periodic table, displays a full inner shell of two electrons, with three electrons in the outermost shell, giving the atom three valence electrons available for bonding. The nucleus consists of 5 protons (red) and 6 neutrons (blue).. You can use craft cotton (or styrofoam tm) balls, poster board, compass, glue, and string to make a model of a boron atom.

The fifth element in the periodic table is the boron(b)... The fifth element in the periodic table is the boron(b). Hto answer this question it is better to understand a little of. So, just represent these 3 valence electrons around the boron atom as a dot. This article gives an idea about the electron configuration of boron(b) and orbital diagram, period and groups, valency and valence electrons of boron, application of different principles. The nucleus consists of 5 protons (red) and 6 neutrons (orange). The nucleus consists of 5 protons (red) and 6 neutrons (orange).

Lewis electron dot diagrams for ions have fewer (for cations) or more (for anions. An orbital diagram is similar to electron configuration, except that instead of indicating the atoms by total numbers, each orbital is shown with up and down. The nucleus consists of 5 protons (red) and 6 neutrons (blue). The fifth element in the periodic table is the boron(b). As, from the bohr diagram of boron, we got to know, it has 3 valence electrons. The bohr diagram for boron shows a central nucleus containing five protons... So, just represent these 3 valence electrons around the boron atom as a dot.

As, from the bohr diagram of boron, we got to know, it has 3 valence electrons. The nucleus consists of 5 protons (red) and 6 neutrons (orange). You can use craft cotton (or styrofoam tm) balls, poster board, compass, glue, and string to make a model of a boron atom. Hto answer this question it is better to understand a little of. And this lewis structure has no resonance and makes a polar molecule with a shape of trigonal planar, with an angle of degrees.. The nucleus consists of 5 protons (red) and 6 neutrons (orange).

The nucleus consists of 5 protons (red) and 6 neutrons (orange)... The fifth element in the periodic table is the boron(b). However, unlike aluminum, it cannot donate. The bohr diagram for boron shows a central nucleus containing five protons. In writing the electron configuration for boron the first two electrons will go in the 1s orbital.

The atomic structure of boron, element number 5 in the periodic table, displays a full inner shell of two electrons, with three electrons in the outermost shell, giving the atom three valence electrons available for bonding. Lewis electron dot diagrams for ions have fewer (for cations) or more (for anions. 5), the most common isotope of the element boron. In writing the electron configuration for boron the first two electrons will go in the 1s orbital... The bohr diagram for boron shows a central nucleus containing five protons.

The nucleus consists of 5 protons (red) and 6 neutrons (blue). An orbital diagram is similar to electron configuration, except that instead of indicating the atoms by total numbers, each orbital is shown with up and down. The nucleus consists of 5 protons (red) and 6 neutrons (blue). 5), the most common isotope of the element boron. A bohr diagram can be used to visually show the bohr model of a particular atom. However, unlike aluminum, it cannot donate. The fifth element in the periodic table is the boron(b).. You can use craft cotton (or styrofoam tm) balls, poster board, compass, glue, and string to make a model of a boron atom.

So, just represent these 3 valence electrons around the boron atom as a dot.. The bohr diagram for boron shows a central nucleus containing five protons. Five electrons (white) occupy available electron shells (rings). In writing the electron configuration for boron the first two electrons will go in the 1s orbital.. Lewis electron dot diagrams for ions have fewer (for cations) or more (for anions.

This article gives an idea about the electron configuration of boron(b) and orbital diagram, period and groups, valency and valence electrons of boron, application of different principles. Hto answer this question it is better to understand a little of. The fifth element in the periodic table is the boron(b). Five electrons (green) bind to the nucleus, successively occupying available electron shells (rings). The bohr diagram for boron shows a central nucleus containing five protons. So, just represent these 3 valence electrons around the boron atom as a dot. In this respect, it resembles aluminum, the next element in the boron group; In writing the electron configuration for boron the first two electrons will go in the 1s orbital.

And this lewis structure has no resonance and makes a polar molecule with a shape of trigonal planar, with an angle of degrees... Lewis electron dot diagrams for ions have fewer (for cations) or more (for anions. As, from the bohr diagram of boron, we got to know, it has 3 valence electrons. A bohr diagram can be used to visually show the bohr model of a particular atom. The nucleus consists of 5 protons (red) and 6 neutrons (orange). Hto answer this question it is better to understand a little of. 5), the most common isotope of the element boron. In this respect, it resembles aluminum, the next element in the boron group; The fifth element in the periodic table is the boron(b). In writing the electron configuration for boron the first two electrons will go in the 1s orbital. The atomic structure of boron, element number 5 in the periodic table, displays a full inner shell of two electrons, with three electrons in the outermost shell, giving the atom three valence electrons available for bonding... The nucleus consists of 5 protons (red) and 6 neutrons (orange).

An orbital diagram is similar to electron configuration, except that instead of indicating the atoms by total numbers, each orbital is shown with up and down.. The fifth element in the periodic table is the boron(b). So, just represent these 3 valence electrons around the boron atom as a dot. This article gives an idea about the electron configuration of boron(b) and orbital diagram, period and groups, valency and valence electrons of boron, application of different principles. The atomic structure of boron, element number 5 in the periodic table, displays a full inner shell of two electrons, with three electrons in the outermost shell, giving the atom three valence electrons available for bonding. Five electrons (green) bind to the nucleus, successively occupying available electron shells (rings). However, unlike aluminum, it cannot donate. In writing the electron configuration for boron the first two electrons will go in the 1s orbital. Five electrons (white) occupy available electron shells (rings).

The nucleus consists of 5 protons (red) and 6 neutrons (blue)... However, unlike aluminum, it cannot donate. The nucleus consists of 5 protons (red) and 6 neutrons (orange). In writing the electron configuration for boron the first two electrons will go in the 1s orbital. The bohr diagram for boron shows a central nucleus containing five protons. 5), the most common isotope of the element boron. The nucleus consists of 5 protons (red) and 6 neutrons (blue). You can use craft cotton (or styrofoam tm) balls, poster board, compass, glue, and string to make a model of a boron atom. Five electrons (white) occupy available electron shells (rings). Electron dot diagram of boron atom. Lewis electron dot diagrams for ions have fewer (for cations) or more (for anions.. 5), the most common isotope of the element boron.

An orbital diagram is similar to electron configuration, except that instead of indicating the atoms by total numbers, each orbital is shown with up and down.. Five electrons (green) bind to the nucleus, successively occupying available electron shells (rings). An orbital diagram is similar to electron configuration, except that instead of indicating the atoms by total numbers, each orbital is shown with up and down. The fifth element in the periodic table is the boron(b).
