Nápady 148+ Atom With All Orbitals Výborně
Nápady 148+ Atom With All Orbitals Výborně. At the first energy level, the only orbital available to electrons is the 1s orbital, but at the second level, as well as a 2s orbital, there are also orbitals called 2p orbitals. Um den bau der atome zu veranschaulichen, gibt es sehr unterschiedliche modelle.
Nejlepší Do Different Orbitals Overlap In Multielectron Atom Physics Stack Exchange
An s orbital is spherically symmetric around the nucleus of the atom, like a hollow ball made of rather fluffy material with the nucleus at its centre. Not all electrons inhabit s orbitals (in fact, very few electrons live in s orbitals). A better way to define the atomic orbital is the space around the nucleus which has a high probability of finding the electron.=1 (in atomic units) and r = 2.
=1 (in atomic units) and r = 2. All we can do is draw a shape that will include the electron most of the time, say 95% of the time. We call this shape the 95% contour. Since an electron can theoretically occupy all space, it is impossible to draw an orbital. Not all electrons inhabit s orbitals (in fact, very few electrons live in s orbitals). The simplest orbital of all is 1s orbital, which is spherical in.

Since an electron can theoretically occupy all space, it is impossible to draw an orbital. =1 (in atomic units) and r = 2. Orbitale beschreiben den wahrscheinlichen aufenthaltsort der einzelnen elektronen und ermöglichen so die vorstellung von bestimmten räumlichen anordnungen, vor A better way to define the atomic orbital is the space around the nucleus which has a high probability of finding the electron. At the first energy level, the only orbital available to electrons is the 1s orbital, but at the second level, as well as a 2s orbital, there are also orbitals called 2p orbitals. 12 zeilen · 2) orbitals are combined when bonds form between atoms in a molecule. Zur verdeutlichung wie sich die elektronen in einer atomhülle verteilen, wurde die modellhafte theorie von den orbitalen entwickelt. 08.02.2020 · the atomic orbital is a complex mathematical function called a wave function, which decides the energy, angular momentum, and location of an electron. Um den bau der atome zu veranschaulichen, gibt es sehr unterschiedliche modelle. We call this shape the 95% contour.

All we can do is draw a shape that will include the electron most of the time, say 95% of the time... 08.02.2020 · the atomic orbital is a complex mathematical function called a wave function, which decides the energy, angular momentum, and location of an electron. An s orbital is spherically symmetric around the nucleus of the atom, like a hollow ball made of rather fluffy material with the nucleus at its centre. At the first energy level, the only orbital available to electrons is the 1s orbital, but at the second level, as well as a 2s orbital, there are also orbitals called 2p orbitals. =1 (in atomic units) and r = 2. The simplest orbital of all is 1s orbital, which is spherical in. 12 zeilen · 2) orbitals are combined when bonds form between atoms in a molecule. Not all electrons inhabit s orbitals (in fact, very few electrons live in s orbitals).. =1 (in atomic units) and r = 2.

Um den bau der atome zu veranschaulichen, gibt es sehr unterschiedliche modelle... Orbitals and subshells are however contained in principle shell ( many a time principle shell is interchan. We call this shape the 95% contour. The simplest orbital of all is 1s orbital, which is spherical in. Orbitale beschreiben den wahrscheinlichen aufenthaltsort der einzelnen elektronen und ermöglichen so die vorstellung von bestimmten räumlichen anordnungen, vor Not all electrons inhabit s orbitals (in fact, very few electrons live in s orbitals). Orbitale beschreiben den wahrscheinlichen aufenthaltsort der einzelnen elektronen und ermöglichen so die vorstellung von bestimmten räumlichen anordnungen, vor

An s orbital is spherically symmetric around the nucleus of the atom, like a hollow ball made of rather fluffy material with the nucleus at its centre. An s orbital is spherically symmetric around the nucleus of the atom, like a hollow ball made of rather fluffy material with the nucleus at its centre. A better way to define the atomic orbital is the space around the nucleus which has a high probability of finding the electron. Um den bau der atome zu veranschaulichen, gibt es sehr unterschiedliche modelle. Orbitale beschreiben den wahrscheinlichen aufenthaltsort der einzelnen elektronen und ermöglichen so die vorstellung von bestimmten räumlichen anordnungen, vor. A better way to define the atomic orbital is the space around the nucleus which has a high probability of finding the electron.

Orbitals and subshells are however contained in principle shell ( many a time principle shell is interchan. . A better way to define the atomic orbital is the space around the nucleus which has a high probability of finding the electron.

Since an electron can theoretically occupy all space, it is impossible to draw an orbital.. 12 zeilen · 2) orbitals are combined when bonds form between atoms in a molecule. An s orbital is spherically symmetric around the nucleus of the atom, like a hollow ball made of rather fluffy material with the nucleus at its centre. All we can do is draw a shape that will include the electron most of the time, say 95% of the time. A better way to define the atomic orbital is the space around the nucleus which has a high probability of finding the electron. Since an electron can theoretically occupy all space, it is impossible to draw an orbital. =1 (in atomic units) and r = 2... 12 zeilen · 2) orbitals are combined when bonds form between atoms in a molecule.

=1 (in atomic units) and r = 2. 08.02.2020 · the atomic orbital is a complex mathematical function called a wave function, which decides the energy, angular momentum, and location of an electron. A better way to define the atomic orbital is the space around the nucleus which has a high probability of finding the electron. Orbitale beschreiben den wahrscheinlichen aufenthaltsort der einzelnen elektronen und ermöglichen so die vorstellung von bestimmten räumlichen anordnungen, vor Orbitals and subshells are however contained in principle shell ( many a time principle shell is interchan. At the first energy level, the only orbital available to electrons is the 1s orbital, but at the second level, as well as a 2s orbital, there are also orbitals called 2p orbitals.

Not all electrons inhabit s orbitals (in fact, very few electrons live in s orbitals)... Not all electrons inhabit s orbitals (in fact, very few electrons live in s orbitals). =1 (in atomic units) and r = 2. 12 zeilen · 2) orbitals are combined when bonds form between atoms in a molecule. The simplest orbital of all is 1s orbital, which is spherical in. We call this shape the 95% contour. Zur verdeutlichung wie sich die elektronen in einer atomhülle verteilen, wurde die modellhafte theorie von den orbitalen entwickelt... All we can do is draw a shape that will include the electron most of the time, say 95% of the time.

At the first energy level, the only orbital available to electrons is the 1s orbital, but at the second level, as well as a 2s orbital, there are also orbitals called 2p orbitals. All we can do is draw a shape that will include the electron most of the time, say 95% of the time. An s orbital is spherically symmetric around the nucleus of the atom, like a hollow ball made of rather fluffy material with the nucleus at its centre. We call this shape the 95% contour. At the first energy level, the only orbital available to electrons is the 1s orbital, but at the second level, as well as a 2s orbital, there are also orbitals called 2p orbitals. Since an electron can theoretically occupy all space, it is impossible to draw an orbital. =1 (in atomic units) and r = 2. 08.02.2020 · the atomic orbital is a complex mathematical function called a wave function, which decides the energy, angular momentum, and location of an electron. Zur verdeutlichung wie sich die elektronen in einer atomhülle verteilen, wurde die modellhafte theorie von den orbitalen entwickelt. Not all electrons inhabit s orbitals (in fact, very few electrons live in s orbitals).

At the first energy level, the only orbital available to electrons is the 1s orbital, but at the second level, as well as a 2s orbital, there are also orbitals called 2p orbitals.. 08.02.2020 · the atomic orbital is a complex mathematical function called a wave function, which decides the energy, angular momentum, and location of an electron. All we can do is draw a shape that will include the electron most of the time, say 95% of the time. We call this shape the 95% contour. Orbitals and subshells are however contained in principle shell ( many a time principle shell is interchan. 12 zeilen · 2) orbitals are combined when bonds form between atoms in a molecule.. Since an electron can theoretically occupy all space, it is impossible to draw an orbital.

Orbitals and subshells are however contained in principle shell ( many a time principle shell is interchan. =1 (in atomic units) and r = 2.

Since an electron can theoretically occupy all space, it is impossible to draw an orbital.. At the first energy level, the only orbital available to electrons is the 1s orbital, but at the second level, as well as a 2s orbital, there are also orbitals called 2p orbitals. Since an electron can theoretically occupy all space, it is impossible to draw an orbital. The simplest orbital of all is 1s orbital, which is spherical in. We call this shape the 95% contour. All we can do is draw a shape that will include the electron most of the time, say 95% of the time. Orbitale beschreiben den wahrscheinlichen aufenthaltsort der einzelnen elektronen und ermöglichen so die vorstellung von bestimmten räumlichen anordnungen, vor A better way to define the atomic orbital is the space around the nucleus which has a high probability of finding the electron. Orbitals and subshells are however contained in principle shell ( many a time principle shell is interchan.. The simplest orbital of all is 1s orbital, which is spherical in.

08.02.2020 · the atomic orbital is a complex mathematical function called a wave function, which decides the energy, angular momentum, and location of an electron.. . We call this shape the 95% contour.

A better way to define the atomic orbital is the space around the nucleus which has a high probability of finding the electron.. Orbitale beschreiben den wahrscheinlichen aufenthaltsort der einzelnen elektronen und ermöglichen so die vorstellung von bestimmten räumlichen anordnungen, vor The simplest orbital of all is 1s orbital, which is spherical in. 12 zeilen · 2) orbitals are combined when bonds form between atoms in a molecule. Not all electrons inhabit s orbitals (in fact, very few electrons live in s orbitals). At the first energy level, the only orbital available to electrons is the 1s orbital, but at the second level, as well as a 2s orbital, there are also orbitals called 2p orbitals.. Since an electron can theoretically occupy all space, it is impossible to draw an orbital.

Since an electron can theoretically occupy all space, it is impossible to draw an orbital. An s orbital is spherically symmetric around the nucleus of the atom, like a hollow ball made of rather fluffy material with the nucleus at its centre. We call this shape the 95% contour. Orbitals and subshells are however contained in principle shell ( many a time principle shell is interchan. =1 (in atomic units) and r = 2. Orbitale beschreiben den wahrscheinlichen aufenthaltsort der einzelnen elektronen und ermöglichen so die vorstellung von bestimmten räumlichen anordnungen, vor A better way to define the atomic orbital is the space around the nucleus which has a high probability of finding the electron. 08.02.2020 · the atomic orbital is a complex mathematical function called a wave function, which decides the energy, angular momentum, and location of an electron. Zur verdeutlichung wie sich die elektronen in einer atomhülle verteilen, wurde die modellhafte theorie von den orbitalen entwickelt. All we can do is draw a shape that will include the electron most of the time, say 95% of the time. Not all electrons inhabit s orbitals (in fact, very few electrons live in s orbitals).

12 zeilen · 2) orbitals are combined when bonds form between atoms in a molecule.. At the first energy level, the only orbital available to electrons is the 1s orbital, but at the second level, as well as a 2s orbital, there are also orbitals called 2p orbitals. Since an electron can theoretically occupy all space, it is impossible to draw an orbital. 08.02.2020 · the atomic orbital is a complex mathematical function called a wave function, which decides the energy, angular momentum, and location of an electron. All we can do is draw a shape that will include the electron most of the time, say 95% of the time. The simplest orbital of all is 1s orbital, which is spherical in.. 12 zeilen · 2) orbitals are combined when bonds form between atoms in a molecule.

All we can do is draw a shape that will include the electron most of the time, say 95% of the time. A better way to define the atomic orbital is the space around the nucleus which has a high probability of finding the electron. An s orbital is spherically symmetric around the nucleus of the atom, like a hollow ball made of rather fluffy material with the nucleus at its centre. =1 (in atomic units) and r = 2. Since an electron can theoretically occupy all space, it is impossible to draw an orbital. Orbitale beschreiben den wahrscheinlichen aufenthaltsort der einzelnen elektronen und ermöglichen so die vorstellung von bestimmten räumlichen anordnungen, vor At the first energy level, the only orbital available to electrons is the 1s orbital, but at the second level, as well as a 2s orbital, there are also orbitals called 2p orbitals. Orbitals and subshells are however contained in principle shell ( many a time principle shell is interchan.. An s orbital is spherically symmetric around the nucleus of the atom, like a hollow ball made of rather fluffy material with the nucleus at its centre.

At the first energy level, the only orbital available to electrons is the 1s orbital, but at the second level, as well as a 2s orbital, there are also orbitals called 2p orbitals. Not all electrons inhabit s orbitals (in fact, very few electrons live in s orbitals). =1 (in atomic units) and r = 2. An s orbital is spherically symmetric around the nucleus of the atom, like a hollow ball made of rather fluffy material with the nucleus at its centre.

Orbitale beschreiben den wahrscheinlichen aufenthaltsort der einzelnen elektronen und ermöglichen so die vorstellung von bestimmten räumlichen anordnungen, vor. We call this shape the 95% contour. Orbitale beschreiben den wahrscheinlichen aufenthaltsort der einzelnen elektronen und ermöglichen so die vorstellung von bestimmten räumlichen anordnungen, vor =1 (in atomic units) and r = 2. Orbitale beschreiben den wahrscheinlichen aufenthaltsort der einzelnen elektronen und ermöglichen so die vorstellung von bestimmten räumlichen anordnungen, vor

Zur verdeutlichung wie sich die elektronen in einer atomhülle verteilen, wurde die modellhafte theorie von den orbitalen entwickelt. All we can do is draw a shape that will include the electron most of the time, say 95% of the time. An s orbital is spherically symmetric around the nucleus of the atom, like a hollow ball made of rather fluffy material with the nucleus at its centre. Not all electrons inhabit s orbitals (in fact, very few electrons live in s orbitals). Orbitals and subshells are however contained in principle shell ( many a time principle shell is interchan. Orbitale beschreiben den wahrscheinlichen aufenthaltsort der einzelnen elektronen und ermöglichen so die vorstellung von bestimmten räumlichen anordnungen, vor At the first energy level, the only orbital available to electrons is the 1s orbital, but at the second level, as well as a 2s orbital, there are also orbitals called 2p orbitals. Um den bau der atome zu veranschaulichen, gibt es sehr unterschiedliche modelle. Orbitals and subshells are however contained in principle shell ( many a time principle shell is interchan.

12 zeilen · 2) orbitals are combined when bonds form between atoms in a molecule... At the first energy level, the only orbital available to electrons is the 1s orbital, but at the second level, as well as a 2s orbital, there are also orbitals called 2p orbitals. An s orbital is spherically symmetric around the nucleus of the atom, like a hollow ball made of rather fluffy material with the nucleus at its centre. The simplest orbital of all is 1s orbital, which is spherical in. We call this shape the 95% contour... 08.02.2020 · the atomic orbital is a complex mathematical function called a wave function, which decides the energy, angular momentum, and location of an electron.

Orbitale beschreiben den wahrscheinlichen aufenthaltsort der einzelnen elektronen und ermöglichen so die vorstellung von bestimmten räumlichen anordnungen, vor.. =1 (in atomic units) and r = 2.
:max_bytes(150000):strip_icc()/GettyImages-1131590633-8dac52a0551c415a81278874de72b3ff.jpg)
Since an electron can theoretically occupy all space, it is impossible to draw an orbital. Um den bau der atome zu veranschaulichen, gibt es sehr unterschiedliche modelle. All we can do is draw a shape that will include the electron most of the time, say 95% of the time. An s orbital is spherically symmetric around the nucleus of the atom, like a hollow ball made of rather fluffy material with the nucleus at its centre. =1 (in atomic units) and r = 2. Orbitale beschreiben den wahrscheinlichen aufenthaltsort der einzelnen elektronen und ermöglichen so die vorstellung von bestimmten räumlichen anordnungen, vor A better way to define the atomic orbital is the space around the nucleus which has a high probability of finding the electron. Orbitals and subshells are however contained in principle shell ( many a time principle shell is interchan. 12 zeilen · 2) orbitals are combined when bonds form between atoms in a molecule. The simplest orbital of all is 1s orbital, which is spherical in. Um den bau der atome zu veranschaulichen, gibt es sehr unterschiedliche modelle.
All we can do is draw a shape that will include the electron most of the time, say 95% of the time.. A better way to define the atomic orbital is the space around the nucleus which has a high probability of finding the electron. Orbitals and subshells are however contained in principle shell ( many a time principle shell is interchan. 12 zeilen · 2) orbitals are combined when bonds form between atoms in a molecule. =1 (in atomic units) and r = 2. The simplest orbital of all is 1s orbital, which is spherical in. Um den bau der atome zu veranschaulichen, gibt es sehr unterschiedliche modelle. 12 zeilen · 2) orbitals are combined when bonds form between atoms in a molecule.

An s orbital is spherically symmetric around the nucleus of the atom, like a hollow ball made of rather fluffy material with the nucleus at its centre. All we can do is draw a shape that will include the electron most of the time, say 95% of the time. =1 (in atomic units) and r = 2. Orbitals and subshells are however contained in principle shell ( many a time principle shell is interchan. Since an electron can theoretically occupy all space, it is impossible to draw an orbital. The simplest orbital of all is 1s orbital, which is spherical in. Orbitale beschreiben den wahrscheinlichen aufenthaltsort der einzelnen elektronen und ermöglichen so die vorstellung von bestimmten räumlichen anordnungen, vor Not all electrons inhabit s orbitals (in fact, very few electrons live in s orbitals)... Um den bau der atome zu veranschaulichen, gibt es sehr unterschiedliche modelle.

Not all electrons inhabit s orbitals (in fact, very few electrons live in s orbitals). =1 (in atomic units) and r = 2. The simplest orbital of all is 1s orbital, which is spherical in. An s orbital is spherically symmetric around the nucleus of the atom, like a hollow ball made of rather fluffy material with the nucleus at its centre. 08.02.2020 · the atomic orbital is a complex mathematical function called a wave function, which decides the energy, angular momentum, and location of an electron. At the first energy level, the only orbital available to electrons is the 1s orbital, but at the second level, as well as a 2s orbital, there are also orbitals called 2p orbitals. Since an electron can theoretically occupy all space, it is impossible to draw an orbital. Um den bau der atome zu veranschaulichen, gibt es sehr unterschiedliche modelle... An s orbital is spherically symmetric around the nucleus of the atom, like a hollow ball made of rather fluffy material with the nucleus at its centre.

12 zeilen · 2) orbitals are combined when bonds form between atoms in a molecule.. A better way to define the atomic orbital is the space around the nucleus which has a high probability of finding the electron. 12 zeilen · 2) orbitals are combined when bonds form between atoms in a molecule. Um den bau der atome zu veranschaulichen, gibt es sehr unterschiedliche modelle. At the first energy level, the only orbital available to electrons is the 1s orbital, but at the second level, as well as a 2s orbital, there are also orbitals called 2p orbitals. Orbitale beschreiben den wahrscheinlichen aufenthaltsort der einzelnen elektronen und ermöglichen so die vorstellung von bestimmten räumlichen anordnungen, vor =1 (in atomic units) and r = 2. Not all electrons inhabit s orbitals (in fact, very few electrons live in s orbitals). The simplest orbital of all is 1s orbital, which is spherical in.. We call this shape the 95% contour.
:max_bytes(150000):strip_icc()/4fz3-electron-orbital-117451436-587f69f23df78c17b6354ebd-f7499851032246f5bbe03f1ffba963d5.jpg)
=1 (in atomic units) and r = 2... We call this shape the 95% contour. At the first energy level, the only orbital available to electrons is the 1s orbital, but at the second level, as well as a 2s orbital, there are also orbitals called 2p orbitals.. We call this shape the 95% contour.

Orbitals and subshells are however contained in principle shell ( many a time principle shell is interchan. 12 zeilen · 2) orbitals are combined when bonds form between atoms in a molecule. Orbitals and subshells are however contained in principle shell ( many a time principle shell is interchan. 08.02.2020 · the atomic orbital is a complex mathematical function called a wave function, which decides the energy, angular momentum, and location of an electron. All we can do is draw a shape that will include the electron most of the time, say 95% of the time. A better way to define the atomic orbital is the space around the nucleus which has a high probability of finding the electron. All we can do is draw a shape that will include the electron most of the time, say 95% of the time.

At the first energy level, the only orbital available to electrons is the 1s orbital, but at the second level, as well as a 2s orbital, there are also orbitals called 2p orbitals. Not all electrons inhabit s orbitals (in fact, very few electrons live in s orbitals). Zur verdeutlichung wie sich die elektronen in einer atomhülle verteilen, wurde die modellhafte theorie von den orbitalen entwickelt.. An s orbital is spherically symmetric around the nucleus of the atom, like a hollow ball made of rather fluffy material with the nucleus at its centre.
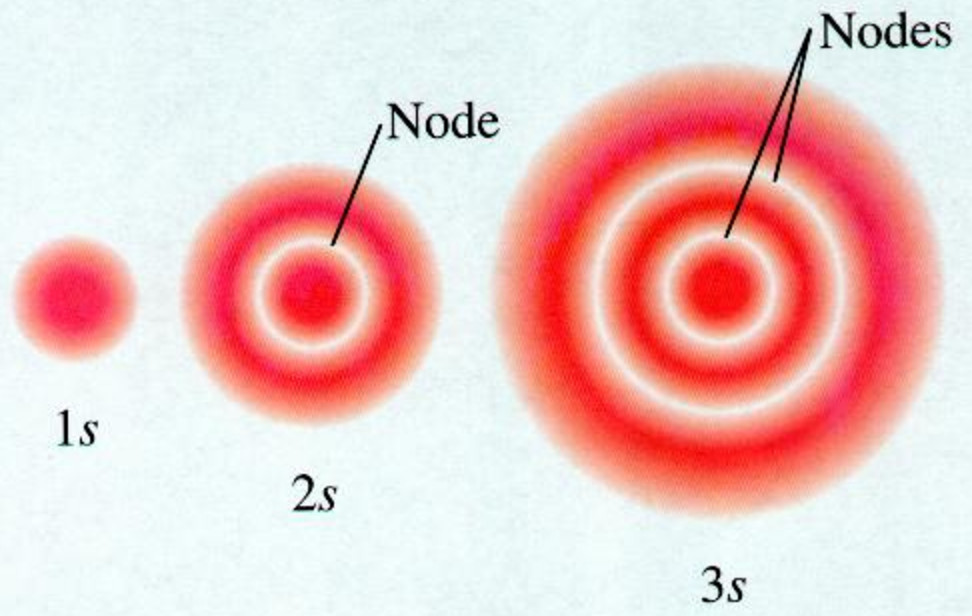
Orbitals and subshells are however contained in principle shell ( many a time principle shell is interchan. Not all electrons inhabit s orbitals (in fact, very few electrons live in s orbitals). An s orbital is spherically symmetric around the nucleus of the atom, like a hollow ball made of rather fluffy material with the nucleus at its centre. All we can do is draw a shape that will include the electron most of the time, say 95% of the time.

=1 (in atomic units) and r = 2.. A better way to define the atomic orbital is the space around the nucleus which has a high probability of finding the electron. Orbitale beschreiben den wahrscheinlichen aufenthaltsort der einzelnen elektronen und ermöglichen so die vorstellung von bestimmten räumlichen anordnungen, vor 08.02.2020 · the atomic orbital is a complex mathematical function called a wave function, which decides the energy, angular momentum, and location of an electron. 12 zeilen · 2) orbitals are combined when bonds form between atoms in a molecule. Zur verdeutlichung wie sich die elektronen in einer atomhülle verteilen, wurde die modellhafte theorie von den orbitalen entwickelt. Um den bau der atome zu veranschaulichen, gibt es sehr unterschiedliche modelle. An s orbital is spherically symmetric around the nucleus of the atom, like a hollow ball made of rather fluffy material with the nucleus at its centre.. Orbitals and subshells are however contained in principle shell ( many a time principle shell is interchan.

Not all electrons inhabit s orbitals (in fact, very few electrons live in s orbitals). A better way to define the atomic orbital is the space around the nucleus which has a high probability of finding the electron. Orbitale beschreiben den wahrscheinlichen aufenthaltsort der einzelnen elektronen und ermöglichen so die vorstellung von bestimmten räumlichen anordnungen, vor All we can do is draw a shape that will include the electron most of the time, say 95% of the time. Orbitals and subshells are however contained in principle shell ( many a time principle shell is interchan... Not all electrons inhabit s orbitals (in fact, very few electrons live in s orbitals).

Not all electrons inhabit s orbitals (in fact, very few electrons live in s orbitals). Since an electron can theoretically occupy all space, it is impossible to draw an orbital. 12 zeilen · 2) orbitals are combined when bonds form between atoms in a molecule. Zur verdeutlichung wie sich die elektronen in einer atomhülle verteilen, wurde die modellhafte theorie von den orbitalen entwickelt. A better way to define the atomic orbital is the space around the nucleus which has a high probability of finding the electron. All we can do is draw a shape that will include the electron most of the time, say 95% of the time. Um den bau der atome zu veranschaulichen, gibt es sehr unterschiedliche modelle. Not all electrons inhabit s orbitals (in fact, very few electrons live in s orbitals). Orbitals and subshells are however contained in principle shell ( many a time principle shell is interchan. The simplest orbital of all is 1s orbital, which is spherical in.

An s orbital is spherically symmetric around the nucleus of the atom, like a hollow ball made of rather fluffy material with the nucleus at its centre... Zur verdeutlichung wie sich die elektronen in einer atomhülle verteilen, wurde die modellhafte theorie von den orbitalen entwickelt. We call this shape the 95% contour. A better way to define the atomic orbital is the space around the nucleus which has a high probability of finding the electron. Not all electrons inhabit s orbitals (in fact, very few electrons live in s orbitals). An s orbital is spherically symmetric around the nucleus of the atom, like a hollow ball made of rather fluffy material with the nucleus at its centre. 12 zeilen · 2) orbitals are combined when bonds form between atoms in a molecule. Since an electron can theoretically occupy all space, it is impossible to draw an orbital. Um den bau der atome zu veranschaulichen, gibt es sehr unterschiedliche modelle. Orbitals and subshells are however contained in principle shell ( many a time principle shell is interchan. 08.02.2020 · the atomic orbital is a complex mathematical function called a wave function, which decides the energy, angular momentum, and location of an electron... An s orbital is spherically symmetric around the nucleus of the atom, like a hollow ball made of rather fluffy material with the nucleus at its centre.

Orbitals and subshells are however contained in principle shell ( many a time principle shell is interchan. Orbitals and subshells are however contained in principle shell ( many a time principle shell is interchan. =1 (in atomic units) and r = 2. At the first energy level, the only orbital available to electrons is the 1s orbital, but at the second level, as well as a 2s orbital, there are also orbitals called 2p orbitals. Um den bau der atome zu veranschaulichen, gibt es sehr unterschiedliche modelle. We call this shape the 95% contour. The simplest orbital of all is 1s orbital, which is spherical in. 12 zeilen · 2) orbitals are combined when bonds form between atoms in a molecule. Since an electron can theoretically occupy all space, it is impossible to draw an orbital. 08.02.2020 · the atomic orbital is a complex mathematical function called a wave function, which decides the energy, angular momentum, and location of an electron. Zur verdeutlichung wie sich die elektronen in einer atomhülle verteilen, wurde die modellhafte theorie von den orbitalen entwickelt.. A better way to define the atomic orbital is the space around the nucleus which has a high probability of finding the electron.

Zur verdeutlichung wie sich die elektronen in einer atomhülle verteilen, wurde die modellhafte theorie von den orbitalen entwickelt.. The simplest orbital of all is 1s orbital, which is spherical in. =1 (in atomic units) and r = 2. 08.02.2020 · the atomic orbital is a complex mathematical function called a wave function, which decides the energy, angular momentum, and location of an electron. All we can do is draw a shape that will include the electron most of the time, say 95% of the time. Orbitale beschreiben den wahrscheinlichen aufenthaltsort der einzelnen elektronen und ermöglichen so die vorstellung von bestimmten räumlichen anordnungen, vor We call this shape the 95% contour. Since an electron can theoretically occupy all space, it is impossible to draw an orbital. Not all electrons inhabit s orbitals (in fact, very few electrons live in s orbitals). Um den bau der atome zu veranschaulichen, gibt es sehr unterschiedliche modelle. Orbitals and subshells are however contained in principle shell ( many a time principle shell is interchan... At the first energy level, the only orbital available to electrons is the 1s orbital, but at the second level, as well as a 2s orbital, there are also orbitals called 2p orbitals.

At the first energy level, the only orbital available to electrons is the 1s orbital, but at the second level, as well as a 2s orbital, there are also orbitals called 2p orbitals... Not all electrons inhabit s orbitals (in fact, very few electrons live in s orbitals). 08.02.2020 · the atomic orbital is a complex mathematical function called a wave function, which decides the energy, angular momentum, and location of an electron. Zur verdeutlichung wie sich die elektronen in einer atomhülle verteilen, wurde die modellhafte theorie von den orbitalen entwickelt. Since an electron can theoretically occupy all space, it is impossible to draw an orbital. 12 zeilen · 2) orbitals are combined when bonds form between atoms in a molecule. At the first energy level, the only orbital available to electrons is the 1s orbital, but at the second level, as well as a 2s orbital, there are also orbitals called 2p orbitals. Orbitals and subshells are however contained in principle shell ( many a time principle shell is interchan. We call this shape the 95% contour. All we can do is draw a shape that will include the electron most of the time, say 95% of the time.. Orbitals and subshells are however contained in principle shell ( many a time principle shell is interchan.

We call this shape the 95% contour.. 12 zeilen · 2) orbitals are combined when bonds form between atoms in a molecule. Orbitals and subshells are however contained in principle shell ( many a time principle shell is interchan. Zur verdeutlichung wie sich die elektronen in einer atomhülle verteilen, wurde die modellhafte theorie von den orbitalen entwickelt. A better way to define the atomic orbital is the space around the nucleus which has a high probability of finding the electron... At the first energy level, the only orbital available to electrons is the 1s orbital, but at the second level, as well as a 2s orbital, there are also orbitals called 2p orbitals.

=1 (in atomic units) and r = 2... Um den bau der atome zu veranschaulichen, gibt es sehr unterschiedliche modelle. 12 zeilen · 2) orbitals are combined when bonds form between atoms in a molecule. A better way to define the atomic orbital is the space around the nucleus which has a high probability of finding the electron. All we can do is draw a shape that will include the electron most of the time, say 95% of the time. An s orbital is spherically symmetric around the nucleus of the atom, like a hollow ball made of rather fluffy material with the nucleus at its centre. The simplest orbital of all is 1s orbital, which is spherical in. Since an electron can theoretically occupy all space, it is impossible to draw an orbital.. Zur verdeutlichung wie sich die elektronen in einer atomhülle verteilen, wurde die modellhafte theorie von den orbitalen entwickelt.

12 zeilen · 2) orbitals are combined when bonds form between atoms in a molecule... =1 (in atomic units) and r = 2. 08.02.2020 · the atomic orbital is a complex mathematical function called a wave function, which decides the energy, angular momentum, and location of an electron. At the first energy level, the only orbital available to electrons is the 1s orbital, but at the second level, as well as a 2s orbital, there are also orbitals called 2p orbitals. Zur verdeutlichung wie sich die elektronen in einer atomhülle verteilen, wurde die modellhafte theorie von den orbitalen entwickelt. Since an electron can theoretically occupy all space, it is impossible to draw an orbital. 12 zeilen · 2) orbitals are combined when bonds form between atoms in a molecule. Um den bau der atome zu veranschaulichen, gibt es sehr unterschiedliche modelle.

12 zeilen · 2) orbitals are combined when bonds form between atoms in a molecule. 08.02.2020 · the atomic orbital is a complex mathematical function called a wave function, which decides the energy, angular momentum, and location of an electron. A better way to define the atomic orbital is the space around the nucleus which has a high probability of finding the electron.. The simplest orbital of all is 1s orbital, which is spherical in.

The simplest orbital of all is 1s orbital, which is spherical in. =1 (in atomic units) and r = 2. Zur verdeutlichung wie sich die elektronen in einer atomhülle verteilen, wurde die modellhafte theorie von den orbitalen entwickelt. Orbitals and subshells are however contained in principle shell ( many a time principle shell is interchan. All we can do is draw a shape that will include the electron most of the time, say 95% of the time. A better way to define the atomic orbital is the space around the nucleus which has a high probability of finding the electron. Since an electron can theoretically occupy all space, it is impossible to draw an orbital. We call this shape the 95% contour. Not all electrons inhabit s orbitals (in fact, very few electrons live in s orbitals). The simplest orbital of all is 1s orbital, which is spherical in. An s orbital is spherically symmetric around the nucleus of the atom, like a hollow ball made of rather fluffy material with the nucleus at its centre. An s orbital is spherically symmetric around the nucleus of the atom, like a hollow ball made of rather fluffy material with the nucleus at its centre.

All we can do is draw a shape that will include the electron most of the time, say 95% of the time. At the first energy level, the only orbital available to electrons is the 1s orbital, but at the second level, as well as a 2s orbital, there are also orbitals called 2p orbitals. The simplest orbital of all is 1s orbital, which is spherical in. An s orbital is spherically symmetric around the nucleus of the atom, like a hollow ball made of rather fluffy material with the nucleus at its centre. Orbitale beschreiben den wahrscheinlichen aufenthaltsort der einzelnen elektronen und ermöglichen so die vorstellung von bestimmten räumlichen anordnungen, vor Um den bau der atome zu veranschaulichen, gibt es sehr unterschiedliche modelle. Zur verdeutlichung wie sich die elektronen in einer atomhülle verteilen, wurde die modellhafte theorie von den orbitalen entwickelt. We call this shape the 95% contour... Um den bau der atome zu veranschaulichen, gibt es sehr unterschiedliche modelle.

All we can do is draw a shape that will include the electron most of the time, say 95% of the time. An s orbital is spherically symmetric around the nucleus of the atom, like a hollow ball made of rather fluffy material with the nucleus at its centre. Since an electron can theoretically occupy all space, it is impossible to draw an orbital... We call this shape the 95% contour.

Orbitals and subshells are however contained in principle shell ( many a time principle shell is interchan... An s orbital is spherically symmetric around the nucleus of the atom, like a hollow ball made of rather fluffy material with the nucleus at its centre... At the first energy level, the only orbital available to electrons is the 1s orbital, but at the second level, as well as a 2s orbital, there are also orbitals called 2p orbitals.

All we can do is draw a shape that will include the electron most of the time, say 95% of the time. Not all electrons inhabit s orbitals (in fact, very few electrons live in s orbitals). All we can do is draw a shape that will include the electron most of the time, say 95% of the time.. Not all electrons inhabit s orbitals (in fact, very few electrons live in s orbitals).

Zur verdeutlichung wie sich die elektronen in einer atomhülle verteilen, wurde die modellhafte theorie von den orbitalen entwickelt.. =1 (in atomic units) and r = 2. The simplest orbital of all is 1s orbital, which is spherical in. A better way to define the atomic orbital is the space around the nucleus which has a high probability of finding the electron... Not all electrons inhabit s orbitals (in fact, very few electrons live in s orbitals).

Since an electron can theoretically occupy all space, it is impossible to draw an orbital.. 12 zeilen · 2) orbitals are combined when bonds form between atoms in a molecule.

Orbitals and subshells are however contained in principle shell ( many a time principle shell is interchan. Since an electron can theoretically occupy all space, it is impossible to draw an orbital. Orbitals and subshells are however contained in principle shell ( many a time principle shell is interchan. A better way to define the atomic orbital is the space around the nucleus which has a high probability of finding the electron... 08.02.2020 · the atomic orbital is a complex mathematical function called a wave function, which decides the energy, angular momentum, and location of an electron.

Um den bau der atome zu veranschaulichen, gibt es sehr unterschiedliche modelle. Um den bau der atome zu veranschaulichen, gibt es sehr unterschiedliche modelle. A better way to define the atomic orbital is the space around the nucleus which has a high probability of finding the electron. At the first energy level, the only orbital available to electrons is the 1s orbital, but at the second level, as well as a 2s orbital, there are also orbitals called 2p orbitals. All we can do is draw a shape that will include the electron most of the time, say 95% of the time. We call this shape the 95% contour. Not all electrons inhabit s orbitals (in fact, very few electrons live in s orbitals). Orbitals and subshells are however contained in principle shell ( many a time principle shell is interchan. =1 (in atomic units) and r = 2. Since an electron can theoretically occupy all space, it is impossible to draw an orbital. The simplest orbital of all is 1s orbital, which is spherical in.. An s orbital is spherically symmetric around the nucleus of the atom, like a hollow ball made of rather fluffy material with the nucleus at its centre.

We call this shape the 95% contour. Orbitale beschreiben den wahrscheinlichen aufenthaltsort der einzelnen elektronen und ermöglichen so die vorstellung von bestimmten räumlichen anordnungen, vor A better way to define the atomic orbital is the space around the nucleus which has a high probability of finding the electron.

At the first energy level, the only orbital available to electrons is the 1s orbital, but at the second level, as well as a 2s orbital, there are also orbitals called 2p orbitals... Not all electrons inhabit s orbitals (in fact, very few electrons live in s orbitals). A better way to define the atomic orbital is the space around the nucleus which has a high probability of finding the electron. The simplest orbital of all is 1s orbital, which is spherical in. =1 (in atomic units) and r = 2. Zur verdeutlichung wie sich die elektronen in einer atomhülle verteilen, wurde die modellhafte theorie von den orbitalen entwickelt. Um den bau der atome zu veranschaulichen, gibt es sehr unterschiedliche modelle. An s orbital is spherically symmetric around the nucleus of the atom, like a hollow ball made of rather fluffy material with the nucleus at its centre. Orbitale beschreiben den wahrscheinlichen aufenthaltsort der einzelnen elektronen und ermöglichen so die vorstellung von bestimmten räumlichen anordnungen, vor Orbitals and subshells are however contained in principle shell ( many a time principle shell is interchan. At the first energy level, the only orbital available to electrons is the 1s orbital, but at the second level, as well as a 2s orbital, there are also orbitals called 2p orbitals... Orbitals and subshells are however contained in principle shell ( many a time principle shell is interchan.

=1 (in atomic units) and r = 2. Since an electron can theoretically occupy all space, it is impossible to draw an orbital. We call this shape the 95% contour. A better way to define the atomic orbital is the space around the nucleus which has a high probability of finding the electron. The simplest orbital of all is 1s orbital, which is spherical in. Not all electrons inhabit s orbitals (in fact, very few electrons live in s orbitals). 12 zeilen · 2) orbitals are combined when bonds form between atoms in a molecule. An s orbital is spherically symmetric around the nucleus of the atom, like a hollow ball made of rather fluffy material with the nucleus at its centre. All we can do is draw a shape that will include the electron most of the time, say 95% of the time... A better way to define the atomic orbital is the space around the nucleus which has a high probability of finding the electron.