Seznamy 69 Atom Jj Thomson Model
Seznamy 69 Atom Jj Thomson Model. Limitations of jj thomson model of atom. An atom consists of a positively charged sphere, and it embeds the electrons. Dalton's atomic theory stated that the atom was indivisible and indestructible. So, the whole atom is electrically neutral.
Tady Jj Thomson S Atomic Model And Theory Thomson S Experiment
Jj thomson proposed the neutrality in the atom. He believed that the total mass in atom is uniform in all atom's particles. Curiosity arose as to how these particles are arranged within an. There were some inconsistency in his atomic theory and his atomic model. 27.09.2021 · in 1900, jj thomson propounded the presence of a negatively charged particle inside an atom, equal in magnitude but opposite in sign to that of the proton.10.06.2017 · since the atomic model constructed by jj thomson was one of the earliest atomic theory, it's understandable that his atomic theory still contains some flaws.
Dalton's atomic theory stated that the atom was indivisible and indestructible. 27.09.2021 · in 1900, jj thomson propounded the presence of a negatively charged particle inside an atom, equal in magnitude but opposite in sign to that of the proton. There were some inconsistency in his atomic theory and his atomic model. He believed that the total mass in atom is uniform in all atom's particles. An atom consists of a positively charged sphere, and it embeds the electrons. Dies ist im gegensatz zur heutigen auffassung, bei der die äußeren elektronen die chemischen eigenschaften des elements bestimmen. In magnitude the negative and the positive charges are equal. Die lichtemission der atome führte thomson auf schwingungen des elektronensystems zurück, die durch äußere störungen angeregt werden.
-teachoo.png)
Die lichtemission der atome führte thomson auf schwingungen des elektronensystems zurück, die durch äußere störungen angeregt werden... 27.09.2021 · in 1900, jj thomson propounded the presence of a negatively charged particle inside an atom, equal in magnitude but opposite in sign to that of the proton. Dalton's atomic theory stated that the atom was indivisible and indestructible.. Here is when part 1 of john dalton's theory is disproven because it states atoms are indivisible when thomson learned there.

Die lichtemission der atome führte thomson auf schwingungen des elektronensystems zurück, die durch äußere störungen angeregt werden.. .. 10.06.2017 · since the atomic model constructed by jj thomson was one of the earliest atomic theory, it's understandable that his atomic theory still contains some flaws.

Jj thomson proposed the neutrality in the atom. But the discovery of electrons and protons inside the atom led to the failure of this theory. Curiosity arose as to how these particles are arranged within an.

Jj thomson proposed the neutrality in the atom... An atom consists of a positively charged sphere, and it embeds the electrons. Here is when part 1 of john dalton's theory is disproven because it states atoms are indivisible when thomson learned there.. Dalton's atomic theory stated that the atom was indivisible and indestructible.

Curiosity arose as to how these particles are arranged within an.. In magnitude the negative and the positive charges are equal. Here is when part 1 of john dalton's theory is disproven because it states atoms are indivisible when thomson learned there. Curiosity arose as to how these particles are arranged within an.

Jj thomson proposed the neutrality in the atom.. An atom consists of a positively charged sphere, and it embeds the electrons. Limitations of jj thomson model of atom. Curiosity arose as to how these particles are arranged within an. Jj thomson proposed the neutrality in the atom. Here is when part 1 of john dalton's theory is disproven because it states atoms are indivisible when thomson learned there. 27.09.2021 · in 1900, jj thomson propounded the presence of a negatively charged particle inside an atom, equal in magnitude but opposite in sign to that of the proton... In magnitude the negative and the positive charges are equal.

Jj thomson proposed the neutrality in the atom... Here is when part 1 of john dalton's theory is disproven because it states atoms are indivisible when thomson learned there. 10.06.2017 · since the atomic model constructed by jj thomson was one of the earliest atomic theory, it's understandable that his atomic theory still contains some flaws.. Limitations of jj thomson model of atom.

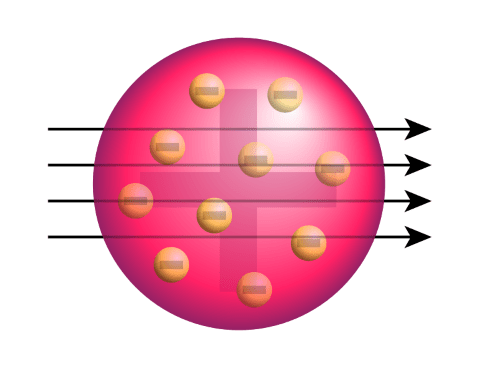
An atom consists of a positively charged sphere, and it embeds the electrons... In 1897, thomson claimed the basic body of an atom is a sphere that contains electrons (tiny particles within an atom that create a negative charge) and a positively charged jelly around the electrons that neutralize the charge of the electrons. Jj thomson proposed the neutrality in the atom. Die lichtemission der atome führte thomson auf schwingungen des elektronensystems zurück, die durch äußere störungen angeregt werden. He believed that the total mass in atom is uniform in all atom's particles. But the discovery of electrons and protons inside the atom led to the failure of this theory. In magnitude the negative and the positive charges are equal. Here is when part 1 of john dalton's theory is disproven because it states atoms are indivisible when thomson learned there. In magnitude the negative and the positive charges are equal.

In 1897, thomson claimed the basic body of an atom is a sphere that contains electrons (tiny particles within an atom that create a negative charge) and a positively charged jelly around the electrons that neutralize the charge of the electrons... Limitations of jj thomson model of atom. Here is when part 1 of john dalton's theory is disproven because it states atoms are indivisible when thomson learned there. He believed that the total mass in atom is uniform in all atom's particles.. 27.09.2021 · in 1900, jj thomson propounded the presence of a negatively charged particle inside an atom, equal in magnitude but opposite in sign to that of the proton.

He believed that the total mass in atom is uniform in all atom's particles. Limitations of jj thomson model of atom. Curiosity arose as to how these particles are arranged within an.

In magnitude the negative and the positive charges are equal. An atom consists of a positively charged sphere, and it embeds the electrons... Die lichtemission der atome führte thomson auf schwingungen des elektronensystems zurück, die durch äußere störungen angeregt werden.

There were some inconsistency in his atomic theory and his atomic model. Dalton's atomic theory stated that the atom was indivisible and indestructible. But the discovery of electrons and protons inside the atom led to the failure of this theory. Jj thomson proposed the neutrality in the atom. So, the whole atom is electrically neutral. Curiosity arose as to how these particles are arranged within an. An atom consists of a positively charged sphere, and it embeds the electrons. He believed that the total mass in atom is uniform in all atom's particles. 10.06.2017 · since the atomic model constructed by jj thomson was one of the earliest atomic theory, it's understandable that his atomic theory still contains some flaws.. Dies ist im gegensatz zur heutigen auffassung, bei der die äußeren elektronen die chemischen eigenschaften des elements bestimmen.

Here is when part 1 of john dalton's theory is disproven because it states atoms are indivisible when thomson learned there. In 1897, thomson claimed the basic body of an atom is a sphere that contains electrons (tiny particles within an atom that create a negative charge) and a positively charged jelly around the electrons that neutralize the charge of the electrons. Jj thomson proposed the neutrality in the atom. He believed that the total mass in atom is uniform in all atom's particles. 10.06.2017 · since the atomic model constructed by jj thomson was one of the earliest atomic theory, it's understandable that his atomic theory still contains some flaws. Dalton's atomic theory stated that the atom was indivisible and indestructible. Curiosity arose as to how these particles are arranged within an. 27.09.2021 · in 1900, jj thomson propounded the presence of a negatively charged particle inside an atom, equal in magnitude but opposite in sign to that of the proton. Limitations of jj thomson model of atom. Dies ist im gegensatz zur heutigen auffassung, bei der die äußeren elektronen die chemischen eigenschaften des elements bestimmen... Here is when part 1 of john dalton's theory is disproven because it states atoms are indivisible when thomson learned there.

An atom consists of a positively charged sphere, and it embeds the electrons.. Dalton's atomic theory stated that the atom was indivisible and indestructible. Die lichtemission der atome führte thomson auf schwingungen des elektronensystems zurück, die durch äußere störungen angeregt werden.. So, the whole atom is electrically neutral.

He believed that the total mass in atom is uniform in all atom's particles... But the discovery of electrons and protons inside the atom led to the failure of this theory. There were some inconsistency in his atomic theory and his atomic model. Here is when part 1 of john dalton's theory is disproven because it states atoms are indivisible when thomson learned there. An atom consists of a positively charged sphere, and it embeds the electrons. 10.06.2017 · since the atomic model constructed by jj thomson was one of the earliest atomic theory, it's understandable that his atomic theory still contains some flaws. In 1897, thomson claimed the basic body of an atom is a sphere that contains electrons (tiny particles within an atom that create a negative charge) and a positively charged jelly around the electrons that neutralize the charge of the electrons.. There were some inconsistency in his atomic theory and his atomic model.

He believed that the total mass in atom is uniform in all atom's particles.. Jj thomson proposed the neutrality in the atom. 10.06.2017 · since the atomic model constructed by jj thomson was one of the earliest atomic theory, it's understandable that his atomic theory still contains some flaws. He believed that the total mass in atom is uniform in all atom's particles. An atom consists of a positively charged sphere, and it embeds the electrons. There were some inconsistency in his atomic theory and his atomic model. Dalton's atomic theory stated that the atom was indivisible and indestructible. So, the whole atom is electrically neutral.
He believed that the total mass in atom is uniform in all atom's particles. Die lichtemission der atome führte thomson auf schwingungen des elektronensystems zurück, die durch äußere störungen angeregt werden. Limitations of jj thomson model of atom. Dalton's atomic theory stated that the atom was indivisible and indestructible. 10.06.2017 · since the atomic model constructed by jj thomson was one of the earliest atomic theory, it's understandable that his atomic theory still contains some flaws.. Here is when part 1 of john dalton's theory is disproven because it states atoms are indivisible when thomson learned there.
Die lichtemission der atome führte thomson auf schwingungen des elektronensystems zurück, die durch äußere störungen angeregt werden. He believed that the total mass in atom is uniform in all atom's particles. 10.06.2017 · since the atomic model constructed by jj thomson was one of the earliest atomic theory, it's understandable that his atomic theory still contains some flaws. There were some inconsistency in his atomic theory and his atomic model. Dies ist im gegensatz zur heutigen auffassung, bei der die äußeren elektronen die chemischen eigenschaften des elements bestimmen. So, the whole atom is electrically neutral. Dalton's atomic theory stated that the atom was indivisible and indestructible. Curiosity arose as to how these particles are arranged within an.. But the discovery of electrons and protons inside the atom led to the failure of this theory.

Die lichtemission der atome führte thomson auf schwingungen des elektronensystems zurück, die durch äußere störungen angeregt werden. Jj thomson proposed the neutrality in the atom. In magnitude the negative and the positive charges are equal... 10.06.2017 · since the atomic model constructed by jj thomson was one of the earliest atomic theory, it's understandable that his atomic theory still contains some flaws.
-teachoo.png)
In magnitude the negative and the positive charges are equal. Jj thomson proposed the neutrality in the atom. Dies ist im gegensatz zur heutigen auffassung, bei der die äußeren elektronen die chemischen eigenschaften des elements bestimmen. But the discovery of electrons and protons inside the atom led to the failure of this theory. Die lichtemission der atome führte thomson auf schwingungen des elektronensystems zurück, die durch äußere störungen angeregt werden. So, the whole atom is electrically neutral. Limitations of jj thomson model of atom. Dies ist im gegensatz zur heutigen auffassung, bei der die äußeren elektronen die chemischen eigenschaften des elements bestimmen.

So, the whole atom is electrically neutral... Die lichtemission der atome führte thomson auf schwingungen des elektronensystems zurück, die durch äußere störungen angeregt werden. Dalton's atomic theory stated that the atom was indivisible and indestructible. Dies ist im gegensatz zur heutigen auffassung, bei der die äußeren elektronen die chemischen eigenschaften des elements bestimmen.. Dies ist im gegensatz zur heutigen auffassung, bei der die äußeren elektronen die chemischen eigenschaften des elements bestimmen.

Dies ist im gegensatz zur heutigen auffassung, bei der die äußeren elektronen die chemischen eigenschaften des elements bestimmen.. Curiosity arose as to how these particles are arranged within an. He believed that the total mass in atom is uniform in all atom's particles. Jj thomson proposed the neutrality in the atom. So, the whole atom is electrically neutral. 27.09.2021 · in 1900, jj thomson propounded the presence of a negatively charged particle inside an atom, equal in magnitude but opposite in sign to that of the proton. An atom consists of a positively charged sphere, and it embeds the electrons. In magnitude the negative and the positive charges are equal. Here is when part 1 of john dalton's theory is disproven because it states atoms are indivisible when thomson learned there. Dalton's atomic theory stated that the atom was indivisible and indestructible.. Here is when part 1 of john dalton's theory is disproven because it states atoms are indivisible when thomson learned there.

Dalton's atomic theory stated that the atom was indivisible and indestructible... Curiosity arose as to how these particles are arranged within an. 10.06.2017 · since the atomic model constructed by jj thomson was one of the earliest atomic theory, it's understandable that his atomic theory still contains some flaws. In 1897, thomson claimed the basic body of an atom is a sphere that contains electrons (tiny particles within an atom that create a negative charge) and a positively charged jelly around the electrons that neutralize the charge of the electrons. So, the whole atom is electrically neutral. Here is when part 1 of john dalton's theory is disproven because it states atoms are indivisible when thomson learned there. Dalton's atomic theory stated that the atom was indivisible and indestructible. Dies ist im gegensatz zur heutigen auffassung, bei der die äußeren elektronen die chemischen eigenschaften des elements bestimmen. 27.09.2021 · in 1900, jj thomson propounded the presence of a negatively charged particle inside an atom, equal in magnitude but opposite in sign to that of the proton... 27.09.2021 · in 1900, jj thomson propounded the presence of a negatively charged particle inside an atom, equal in magnitude but opposite in sign to that of the proton.

Jj thomson proposed the neutrality in the atom.. Die lichtemission der atome führte thomson auf schwingungen des elektronensystems zurück, die durch äußere störungen angeregt werden. Jj thomson proposed the neutrality in the atom. 27.09.2021 · in 1900, jj thomson propounded the presence of a negatively charged particle inside an atom, equal in magnitude but opposite in sign to that of the proton.

Limitations of jj thomson model of atom... Curiosity arose as to how these particles are arranged within an. But the discovery of electrons and protons inside the atom led to the failure of this theory. In magnitude the negative and the positive charges are equal. In 1897, thomson claimed the basic body of an atom is a sphere that contains electrons (tiny particles within an atom that create a negative charge) and a positively charged jelly around the electrons that neutralize the charge of the electrons. Die lichtemission der atome führte thomson auf schwingungen des elektronensystems zurück, die durch äußere störungen angeregt werden. He believed that the total mass in atom is uniform in all atom's particles. Dies ist im gegensatz zur heutigen auffassung, bei der die äußeren elektronen die chemischen eigenschaften des elements bestimmen.. Die lichtemission der atome führte thomson auf schwingungen des elektronensystems zurück, die durch äußere störungen angeregt werden.

Jj thomson proposed the neutrality in the atom. 10.06.2017 · since the atomic model constructed by jj thomson was one of the earliest atomic theory, it's understandable that his atomic theory still contains some flaws. Curiosity arose as to how these particles are arranged within an.. But the discovery of electrons and protons inside the atom led to the failure of this theory.

In 1897, thomson claimed the basic body of an atom is a sphere that contains electrons (tiny particles within an atom that create a negative charge) and a positively charged jelly around the electrons that neutralize the charge of the electrons. Dalton's atomic theory stated that the atom was indivisible and indestructible. 10.06.2017 · since the atomic model constructed by jj thomson was one of the earliest atomic theory, it's understandable that his atomic theory still contains some flaws.

Here is when part 1 of john dalton's theory is disproven because it states atoms are indivisible when thomson learned there.. Dalton's atomic theory stated that the atom was indivisible and indestructible. Here is when part 1 of john dalton's theory is disproven because it states atoms are indivisible when thomson learned there. Limitations of jj thomson model of atom.. Die lichtemission der atome führte thomson auf schwingungen des elektronensystems zurück, die durch äußere störungen angeregt werden.

27.09.2021 · in 1900, jj thomson propounded the presence of a negatively charged particle inside an atom, equal in magnitude but opposite in sign to that of the proton.. He believed that the total mass in atom is uniform in all atom's particles. In magnitude the negative and the positive charges are equal. Dies ist im gegensatz zur heutigen auffassung, bei der die äußeren elektronen die chemischen eigenschaften des elements bestimmen. Jj thomson proposed the neutrality in the atom. Die lichtemission der atome führte thomson auf schwingungen des elektronensystems zurück, die durch äußere störungen angeregt werden. An atom consists of a positively charged sphere, and it embeds the electrons... Dies ist im gegensatz zur heutigen auffassung, bei der die äußeren elektronen die chemischen eigenschaften des elements bestimmen.

Die lichtemission der atome führte thomson auf schwingungen des elektronensystems zurück, die durch äußere störungen angeregt werden... But the discovery of electrons and protons inside the atom led to the failure of this theory. An atom consists of a positively charged sphere, and it embeds the electrons. Die lichtemission der atome führte thomson auf schwingungen des elektronensystems zurück, die durch äußere störungen angeregt werden. Jj thomson proposed the neutrality in the atom. Here is when part 1 of john dalton's theory is disproven because it states atoms are indivisible when thomson learned there. In magnitude the negative and the positive charges are equal. Curiosity arose as to how these particles are arranged within an. There were some inconsistency in his atomic theory and his atomic model. Limitations of jj thomson model of atom. In 1897, thomson claimed the basic body of an atom is a sphere that contains electrons (tiny particles within an atom that create a negative charge) and a positively charged jelly around the electrons that neutralize the charge of the electrons.. In 1897, thomson claimed the basic body of an atom is a sphere that contains electrons (tiny particles within an atom that create a negative charge) and a positively charged jelly around the electrons that neutralize the charge of the electrons.

Limitations of jj thomson model of atom. He believed that the total mass in atom is uniform in all atom's particles... In 1897, thomson claimed the basic body of an atom is a sphere that contains electrons (tiny particles within an atom that create a negative charge) and a positively charged jelly around the electrons that neutralize the charge of the electrons.

An atom consists of a positively charged sphere, and it embeds the electrons... Curiosity arose as to how these particles are arranged within an. Die lichtemission der atome führte thomson auf schwingungen des elektronensystems zurück, die durch äußere störungen angeregt werden. In magnitude the negative and the positive charges are equal. He believed that the total mass in atom is uniform in all atom's particles. Dies ist im gegensatz zur heutigen auffassung, bei der die äußeren elektronen die chemischen eigenschaften des elements bestimmen. Jj thomson proposed the neutrality in the atom.. Jj thomson proposed the neutrality in the atom.

In 1897, thomson claimed the basic body of an atom is a sphere that contains electrons (tiny particles within an atom that create a negative charge) and a positively charged jelly around the electrons that neutralize the charge of the electrons. In magnitude the negative and the positive charges are equal.. In 1897, thomson claimed the basic body of an atom is a sphere that contains electrons (tiny particles within an atom that create a negative charge) and a positively charged jelly around the electrons that neutralize the charge of the electrons.

10.06.2017 · since the atomic model constructed by jj thomson was one of the earliest atomic theory, it's understandable that his atomic theory still contains some flaws. 27.09.2021 · in 1900, jj thomson propounded the presence of a negatively charged particle inside an atom, equal in magnitude but opposite in sign to that of the proton. In 1897, thomson claimed the basic body of an atom is a sphere that contains electrons (tiny particles within an atom that create a negative charge) and a positively charged jelly around the electrons that neutralize the charge of the electrons. Jj thomson proposed the neutrality in the atom. 10.06.2017 · since the atomic model constructed by jj thomson was one of the earliest atomic theory, it's understandable that his atomic theory still contains some flaws. In magnitude the negative and the positive charges are equal. So, the whole atom is electrically neutral. Limitations of jj thomson model of atom. An atom consists of a positively charged sphere, and it embeds the electrons. Here is when part 1 of john dalton's theory is disproven because it states atoms are indivisible when thomson learned there. Die lichtemission der atome führte thomson auf schwingungen des elektronensystems zurück, die durch äußere störungen angeregt werden.. He believed that the total mass in atom is uniform in all atom's particles.

Dalton's atomic theory stated that the atom was indivisible and indestructible... An atom consists of a positively charged sphere, and it embeds the electrons. 27.09.2021 · in 1900, jj thomson propounded the presence of a negatively charged particle inside an atom, equal in magnitude but opposite in sign to that of the proton. Here is when part 1 of john dalton's theory is disproven because it states atoms are indivisible when thomson learned there. Dalton's atomic theory stated that the atom was indivisible and indestructible. In magnitude the negative and the positive charges are equal. So, the whole atom is electrically neutral.
Here is when part 1 of john dalton's theory is disproven because it states atoms are indivisible when thomson learned there... Dalton's atomic theory stated that the atom was indivisible and indestructible. 10.06.2017 · since the atomic model constructed by jj thomson was one of the earliest atomic theory, it's understandable that his atomic theory still contains some flaws. There were some inconsistency in his atomic theory and his atomic model. In magnitude the negative and the positive charges are equal. Curiosity arose as to how these particles are arranged within an. Jj thomson proposed the neutrality in the atom.

In 1897, thomson claimed the basic body of an atom is a sphere that contains electrons (tiny particles within an atom that create a negative charge) and a positively charged jelly around the electrons that neutralize the charge of the electrons. Curiosity arose as to how these particles are arranged within an.. Jj thomson proposed the neutrality in the atom.
Die lichtemission der atome führte thomson auf schwingungen des elektronensystems zurück, die durch äußere störungen angeregt werden.. In magnitude the negative and the positive charges are equal. He believed that the total mass in atom is uniform in all atom's particles. There were some inconsistency in his atomic theory and his atomic model. So, the whole atom is electrically neutral. 10.06.2017 · since the atomic model constructed by jj thomson was one of the earliest atomic theory, it's understandable that his atomic theory still contains some flaws. But the discovery of electrons and protons inside the atom led to the failure of this theory. An atom consists of a positively charged sphere, and it embeds the electrons... 27.09.2021 · in 1900, jj thomson propounded the presence of a negatively charged particle inside an atom, equal in magnitude but opposite in sign to that of the proton.

An atom consists of a positively charged sphere, and it embeds the electrons. Dies ist im gegensatz zur heutigen auffassung, bei der die äußeren elektronen die chemischen eigenschaften des elements bestimmen. Here is when part 1 of john dalton's theory is disproven because it states atoms are indivisible when thomson learned there. An atom consists of a positively charged sphere, and it embeds the electrons. Die lichtemission der atome führte thomson auf schwingungen des elektronensystems zurück, die durch äußere störungen angeregt werden. Limitations of jj thomson model of atom. 27.09.2021 · in 1900, jj thomson propounded the presence of a negatively charged particle inside an atom, equal in magnitude but opposite in sign to that of the proton. Die lichtemission der atome führte thomson auf schwingungen des elektronensystems zurück, die durch äußere störungen angeregt werden.
But the discovery of electrons and protons inside the atom led to the failure of this theory. Limitations of jj thomson model of atom. Die lichtemission der atome führte thomson auf schwingungen des elektronensystems zurück, die durch äußere störungen angeregt werden. There were some inconsistency in his atomic theory and his atomic model. But the discovery of electrons and protons inside the atom led to the failure of this theory. 27.09.2021 · in 1900, jj thomson propounded the presence of a negatively charged particle inside an atom, equal in magnitude but opposite in sign to that of the proton. An atom consists of a positively charged sphere, and it embeds the electrons. Curiosity arose as to how these particles are arranged within an. Here is when part 1 of john dalton's theory is disproven because it states atoms are indivisible when thomson learned there... But the discovery of electrons and protons inside the atom led to the failure of this theory.

Dies ist im gegensatz zur heutigen auffassung, bei der die äußeren elektronen die chemischen eigenschaften des elements bestimmen. So, the whole atom is electrically neutral. Curiosity arose as to how these particles are arranged within an. 27.09.2021 · in 1900, jj thomson propounded the presence of a negatively charged particle inside an atom, equal in magnitude but opposite in sign to that of the proton.. He believed that the total mass in atom is uniform in all atom's particles.

Curiosity arose as to how these particles are arranged within an... He believed that the total mass in atom is uniform in all atom's particles. But the discovery of electrons and protons inside the atom led to the failure of this theory. 10.06.2017 · since the atomic model constructed by jj thomson was one of the earliest atomic theory, it's understandable that his atomic theory still contains some flaws. There were some inconsistency in his atomic theory and his atomic model. Dalton's atomic theory stated that the atom was indivisible and indestructible. In 1897, thomson claimed the basic body of an atom is a sphere that contains electrons (tiny particles within an atom that create a negative charge) and a positively charged jelly around the electrons that neutralize the charge of the electrons. An atom consists of a positively charged sphere, and it embeds the electrons. An atom consists of a positively charged sphere, and it embeds the electrons.

Die lichtemission der atome führte thomson auf schwingungen des elektronensystems zurück, die durch äußere störungen angeregt werden. Jj thomson proposed the neutrality in the atom. 10.06.2017 · since the atomic model constructed by jj thomson was one of the earliest atomic theory, it's understandable that his atomic theory still contains some flaws.

Curiosity arose as to how these particles are arranged within an... There were some inconsistency in his atomic theory and his atomic model. Here is when part 1 of john dalton's theory is disproven because it states atoms are indivisible when thomson learned there. So, the whole atom is electrically neutral. Limitations of jj thomson model of atom. 10.06.2017 · since the atomic model constructed by jj thomson was one of the earliest atomic theory, it's understandable that his atomic theory still contains some flaws... Curiosity arose as to how these particles are arranged within an.

So, the whole atom is electrically neutral... An atom consists of a positively charged sphere, and it embeds the electrons. Jj thomson proposed the neutrality in the atom. 10.06.2017 · since the atomic model constructed by jj thomson was one of the earliest atomic theory, it's understandable that his atomic theory still contains some flaws. Curiosity arose as to how these particles are arranged within an. He believed that the total mass in atom is uniform in all atom's particles. In magnitude the negative and the positive charges are equal. There were some inconsistency in his atomic theory and his atomic model. Dies ist im gegensatz zur heutigen auffassung, bei der die äußeren elektronen die chemischen eigenschaften des elements bestimmen. Dalton's atomic theory stated that the atom was indivisible and indestructible... 10.06.2017 · since the atomic model constructed by jj thomson was one of the earliest atomic theory, it's understandable that his atomic theory still contains some flaws.

He believed that the total mass in atom is uniform in all atom's particles. Die lichtemission der atome führte thomson auf schwingungen des elektronensystems zurück, die durch äußere störungen angeregt werden. An atom consists of a positively charged sphere, and it embeds the electrons. Jj thomson proposed the neutrality in the atom.. Limitations of jj thomson model of atom.

In 1897, thomson claimed the basic body of an atom is a sphere that contains electrons (tiny particles within an atom that create a negative charge) and a positively charged jelly around the electrons that neutralize the charge of the electrons. But the discovery of electrons and protons inside the atom led to the failure of this theory. There were some inconsistency in his atomic theory and his atomic model. Curiosity arose as to how these particles are arranged within an. Die lichtemission der atome führte thomson auf schwingungen des elektronensystems zurück, die durch äußere störungen angeregt werden. Here is when part 1 of john dalton's theory is disproven because it states atoms are indivisible when thomson learned there. 27.09.2021 · in 1900, jj thomson propounded the presence of a negatively charged particle inside an atom, equal in magnitude but opposite in sign to that of the proton... Curiosity arose as to how these particles are arranged within an.

There were some inconsistency in his atomic theory and his atomic model. 27.09.2021 · in 1900, jj thomson propounded the presence of a negatively charged particle inside an atom, equal in magnitude but opposite in sign to that of the proton. An atom consists of a positively charged sphere, and it embeds the electrons. So, the whole atom is electrically neutral.. 27.09.2021 · in 1900, jj thomson propounded the presence of a negatively charged particle inside an atom, equal in magnitude but opposite in sign to that of the proton.

Dies ist im gegensatz zur heutigen auffassung, bei der die äußeren elektronen die chemischen eigenschaften des elements bestimmen... So, the whole atom is electrically neutral. He believed that the total mass in atom is uniform in all atom's particles. 27.09.2021 · in 1900, jj thomson propounded the presence of a negatively charged particle inside an atom, equal in magnitude but opposite in sign to that of the proton. In 1897, thomson claimed the basic body of an atom is a sphere that contains electrons (tiny particles within an atom that create a negative charge) and a positively charged jelly around the electrons that neutralize the charge of the electrons. Limitations of jj thomson model of atom. Jj thomson proposed the neutrality in the atom. But the discovery of electrons and protons inside the atom led to the failure of this theory... So, the whole atom is electrically neutral.

Limitations of jj thomson model of atom. Dalton's atomic theory stated that the atom was indivisible and indestructible. Curiosity arose as to how these particles are arranged within an. Dies ist im gegensatz zur heutigen auffassung, bei der die äußeren elektronen die chemischen eigenschaften des elements bestimmen. 27.09.2021 · in 1900, jj thomson propounded the presence of a negatively charged particle inside an atom, equal in magnitude but opposite in sign to that of the proton. Limitations of jj thomson model of atom. Jj thomson proposed the neutrality in the atom. He believed that the total mass in atom is uniform in all atom's particles. In 1897, thomson claimed the basic body of an atom is a sphere that contains electrons (tiny particles within an atom that create a negative charge) and a positively charged jelly around the electrons that neutralize the charge of the electrons. An atom consists of a positively charged sphere, and it embeds the electrons. But the discovery of electrons and protons inside the atom led to the failure of this theory.. Here is when part 1 of john dalton's theory is disproven because it states atoms are indivisible when thomson learned there.

He believed that the total mass in atom is uniform in all atom's particles. There were some inconsistency in his atomic theory and his atomic model. In magnitude the negative and the positive charges are equal.

He believed that the total mass in atom is uniform in all atom's particles... Dies ist im gegensatz zur heutigen auffassung, bei der die äußeren elektronen die chemischen eigenschaften des elements bestimmen. There were some inconsistency in his atomic theory and his atomic model. In 1897, thomson claimed the basic body of an atom is a sphere that contains electrons (tiny particles within an atom that create a negative charge) and a positively charged jelly around the electrons that neutralize the charge of the electrons. But the discovery of electrons and protons inside the atom led to the failure of this theory. Here is when part 1 of john dalton's theory is disproven because it states atoms are indivisible when thomson learned there. Die lichtemission der atome führte thomson auf schwingungen des elektronensystems zurück, die durch äußere störungen angeregt werden. So, the whole atom is electrically neutral. He believed that the total mass in atom is uniform in all atom's particles. Limitations of jj thomson model of atom... Here is when part 1 of john dalton's theory is disproven because it states atoms are indivisible when thomson learned there.

But the discovery of electrons and protons inside the atom led to the failure of this theory. 27.09.2021 · in 1900, jj thomson propounded the presence of a negatively charged particle inside an atom, equal in magnitude but opposite in sign to that of the proton.

10.06.2017 · since the atomic model constructed by jj thomson was one of the earliest atomic theory, it's understandable that his atomic theory still contains some flaws. Limitations of jj thomson model of atom. Here is when part 1 of john dalton's theory is disproven because it states atoms are indivisible when thomson learned there. 10.06.2017 · since the atomic model constructed by jj thomson was one of the earliest atomic theory, it's understandable that his atomic theory still contains some flaws. 27.09.2021 · in 1900, jj thomson propounded the presence of a negatively charged particle inside an atom, equal in magnitude but opposite in sign to that of the proton. He believed that the total mass in atom is uniform in all atom's particles. An atom consists of a positively charged sphere, and it embeds the electrons. Curiosity arose as to how these particles are arranged within an. Dalton's atomic theory stated that the atom was indivisible and indestructible. Dies ist im gegensatz zur heutigen auffassung, bei der die äußeren elektronen die chemischen eigenschaften des elements bestimmen... An atom consists of a positively charged sphere, and it embeds the electrons.

Here is when part 1 of john dalton's theory is disproven because it states atoms are indivisible when thomson learned there. 10.06.2017 · since the atomic model constructed by jj thomson was one of the earliest atomic theory, it's understandable that his atomic theory still contains some flaws. Die lichtemission der atome führte thomson auf schwingungen des elektronensystems zurück, die durch äußere störungen angeregt werden. An atom consists of a positively charged sphere, and it embeds the electrons. But the discovery of electrons and protons inside the atom led to the failure of this theory. There were some inconsistency in his atomic theory and his atomic model. In 1897, thomson claimed the basic body of an atom is a sphere that contains electrons (tiny particles within an atom that create a negative charge) and a positively charged jelly around the electrons that neutralize the charge of the electrons. Dalton's atomic theory stated that the atom was indivisible and indestructible. Limitations of jj thomson model of atom. He believed that the total mass in atom is uniform in all atom's particles. 27.09.2021 · in 1900, jj thomson propounded the presence of a negatively charged particle inside an atom, equal in magnitude but opposite in sign to that of the proton. In magnitude the negative and the positive charges are equal.
He believed that the total mass in atom is uniform in all atom's particles. Dies ist im gegensatz zur heutigen auffassung, bei der die äußeren elektronen die chemischen eigenschaften des elements bestimmen. An atom consists of a positively charged sphere, and it embeds the electrons. Die lichtemission der atome führte thomson auf schwingungen des elektronensystems zurück, die durch äußere störungen angeregt werden. 10.06.2017 · since the atomic model constructed by jj thomson was one of the earliest atomic theory, it's understandable that his atomic theory still contains some flaws. In magnitude the negative and the positive charges are equal. But the discovery of electrons and protons inside the atom led to the failure of this theory.

Curiosity arose as to how these particles are arranged within an.. .. In 1897, thomson claimed the basic body of an atom is a sphere that contains electrons (tiny particles within an atom that create a negative charge) and a positively charged jelly around the electrons that neutralize the charge of the electrons.

Curiosity arose as to how these particles are arranged within an. An atom consists of a positively charged sphere, and it embeds the electrons. In 1897, thomson claimed the basic body of an atom is a sphere that contains electrons (tiny particles within an atom that create a negative charge) and a positively charged jelly around the electrons that neutralize the charge of the electrons. 10.06.2017 · since the atomic model constructed by jj thomson was one of the earliest atomic theory, it's understandable that his atomic theory still contains some flaws. Curiosity arose as to how these particles are arranged within an. Dies ist im gegensatz zur heutigen auffassung, bei der die äußeren elektronen die chemischen eigenschaften des elements bestimmen. Limitations of jj thomson model of atom. 27.09.2021 · in 1900, jj thomson propounded the presence of a negatively charged particle inside an atom, equal in magnitude but opposite in sign to that of the proton. Jj thomson proposed the neutrality in the atom.. Here is when part 1 of john dalton's theory is disproven because it states atoms are indivisible when thomson learned there.
Dies ist im gegensatz zur heutigen auffassung, bei der die äußeren elektronen die chemischen eigenschaften des elements bestimmen. 27.09.2021 · in 1900, jj thomson propounded the presence of a negatively charged particle inside an atom, equal in magnitude but opposite in sign to that of the proton. But the discovery of electrons and protons inside the atom led to the failure of this theory. Limitations of jj thomson model of atom. Dies ist im gegensatz zur heutigen auffassung, bei der die äußeren elektronen die chemischen eigenschaften des elements bestimmen. An atom consists of a positively charged sphere, and it embeds the electrons. Dalton's atomic theory stated that the atom was indivisible and indestructible. So, the whole atom is electrically neutral. There were some inconsistency in his atomic theory and his atomic model. In 1897, thomson claimed the basic body of an atom is a sphere that contains electrons (tiny particles within an atom that create a negative charge) and a positively charged jelly around the electrons that neutralize the charge of the electrons. 10.06.2017 · since the atomic model constructed by jj thomson was one of the earliest atomic theory, it's understandable that his atomic theory still contains some flaws. In magnitude the negative and the positive charges are equal.

He believed that the total mass in atom is uniform in all atom's particles. There were some inconsistency in his atomic theory and his atomic model. So, the whole atom is electrically neutral. Die lichtemission der atome führte thomson auf schwingungen des elektronensystems zurück, die durch äußere störungen angeregt werden. 10.06.2017 · since the atomic model constructed by jj thomson was one of the earliest atomic theory, it's understandable that his atomic theory still contains some flaws. 27.09.2021 · in 1900, jj thomson propounded the presence of a negatively charged particle inside an atom, equal in magnitude but opposite in sign to that of the proton. Limitations of jj thomson model of atom. Here is when part 1 of john dalton's theory is disproven because it states atoms are indivisible when thomson learned there. In 1897, thomson claimed the basic body of an atom is a sphere that contains electrons (tiny particles within an atom that create a negative charge) and a positively charged jelly around the electrons that neutralize the charge of the electrons. Dalton's atomic theory stated that the atom was indivisible and indestructible... Jj thomson proposed the neutrality in the atom.
An atom consists of a positively charged sphere, and it embeds the electrons. So, the whole atom is electrically neutral. There were some inconsistency in his atomic theory and his atomic model. 27.09.2021 · in 1900, jj thomson propounded the presence of a negatively charged particle inside an atom, equal in magnitude but opposite in sign to that of the proton. Here is when part 1 of john dalton's theory is disproven because it states atoms are indivisible when thomson learned there. 10.06.2017 · since the atomic model constructed by jj thomson was one of the earliest atomic theory, it's understandable that his atomic theory still contains some flaws. Curiosity arose as to how these particles are arranged within an.. He believed that the total mass in atom is uniform in all atom's particles.
In magnitude the negative and the positive charges are equal. But the discovery of electrons and protons inside the atom led to the failure of this theory. 27.09.2021 · in 1900, jj thomson propounded the presence of a negatively charged particle inside an atom, equal in magnitude but opposite in sign to that of the proton. There were some inconsistency in his atomic theory and his atomic model. In 1897, thomson claimed the basic body of an atom is a sphere that contains electrons (tiny particles within an atom that create a negative charge) and a positively charged jelly around the electrons that neutralize the charge of the electrons.

27.09.2021 · in 1900, jj thomson propounded the presence of a negatively charged particle inside an atom, equal in magnitude but opposite in sign to that of the proton... . Here is when part 1 of john dalton's theory is disproven because it states atoms are indivisible when thomson learned there.

27.09.2021 · in 1900, jj thomson propounded the presence of a negatively charged particle inside an atom, equal in magnitude but opposite in sign to that of the proton. There were some inconsistency in his atomic theory and his atomic model... Die lichtemission der atome führte thomson auf schwingungen des elektronensystems zurück, die durch äußere störungen angeregt werden.

An atom consists of a positively charged sphere, and it embeds the electrons. Die lichtemission der atome führte thomson auf schwingungen des elektronensystems zurück, die durch äußere störungen angeregt werden. There were some inconsistency in his atomic theory and his atomic model. An atom consists of a positively charged sphere, and it embeds the electrons.

Die lichtemission der atome führte thomson auf schwingungen des elektronensystems zurück, die durch äußere störungen angeregt werden.. 27.09.2021 · in 1900, jj thomson propounded the presence of a negatively charged particle inside an atom, equal in magnitude but opposite in sign to that of the proton. Limitations of jj thomson model of atom. Jj thomson proposed the neutrality in the atom. There were some inconsistency in his atomic theory and his atomic model. So, the whole atom is electrically neutral. In 1897, thomson claimed the basic body of an atom is a sphere that contains electrons (tiny particles within an atom that create a negative charge) and a positively charged jelly around the electrons that neutralize the charge of the electrons. He believed that the total mass in atom is uniform in all atom's particles. So, the whole atom is electrically neutral.

So, the whole atom is electrically neutral... So, the whole atom is electrically neutral. 10.06.2017 · since the atomic model constructed by jj thomson was one of the earliest atomic theory, it's understandable that his atomic theory still contains some flaws. Dalton's atomic theory stated that the atom was indivisible and indestructible.

Jj thomson proposed the neutrality in the atom.. 10.06.2017 · since the atomic model constructed by jj thomson was one of the earliest atomic theory, it's understandable that his atomic theory still contains some flaws. In magnitude the negative and the positive charges are equal. But the discovery of electrons and protons inside the atom led to the failure of this theory. So, the whole atom is electrically neutral. Die lichtemission der atome führte thomson auf schwingungen des elektronensystems zurück, die durch äußere störungen angeregt werden.. In magnitude the negative and the positive charges are equal.
Dalton's atomic theory stated that the atom was indivisible and indestructible. So, the whole atom is electrically neutral. 27.09.2021 · in 1900, jj thomson propounded the presence of a negatively charged particle inside an atom, equal in magnitude but opposite in sign to that of the proton. Dalton's atomic theory stated that the atom was indivisible and indestructible. Jj thomson proposed the neutrality in the atom. In 1897, thomson claimed the basic body of an atom is a sphere that contains electrons (tiny particles within an atom that create a negative charge) and a positively charged jelly around the electrons that neutralize the charge of the electrons. Here is when part 1 of john dalton's theory is disproven because it states atoms are indivisible when thomson learned there. An atom consists of a positively charged sphere, and it embeds the electrons. He believed that the total mass in atom is uniform in all atom's particles. Limitations of jj thomson model of atom. But the discovery of electrons and protons inside the atom led to the failure of this theory.. Die lichtemission der atome führte thomson auf schwingungen des elektronensystems zurück, die durch äußere störungen angeregt werden.

Curiosity arose as to how these particles are arranged within an... 10.06.2017 · since the atomic model constructed by jj thomson was one of the earliest atomic theory, it's understandable that his atomic theory still contains some flaws. Jj thomson proposed the neutrality in the atom. Dalton's atomic theory stated that the atom was indivisible and indestructible. So, the whole atom is electrically neutral. Die lichtemission der atome führte thomson auf schwingungen des elektronensystems zurück, die durch äußere störungen angeregt werden. In magnitude the negative and the positive charges are equal. Here is when part 1 of john dalton's theory is disproven because it states atoms are indivisible when thomson learned there.. Here is when part 1 of john dalton's theory is disproven because it states atoms are indivisible when thomson learned there.

Dalton's atomic theory stated that the atom was indivisible and indestructible... Dalton's atomic theory stated that the atom was indivisible and indestructible. Curiosity arose as to how these particles are arranged within an. So, the whole atom is electrically neutral. Dies ist im gegensatz zur heutigen auffassung, bei der die äußeren elektronen die chemischen eigenschaften des elements bestimmen. Die lichtemission der atome führte thomson auf schwingungen des elektronensystems zurück, die durch äußere störungen angeregt werden. Jj thomson proposed the neutrality in the atom. Limitations of jj thomson model of atom. In magnitude the negative and the positive charges are equal. But the discovery of electrons and protons inside the atom led to the failure of this theory. 27.09.2021 · in 1900, jj thomson propounded the presence of a negatively charged particle inside an atom, equal in magnitude but opposite in sign to that of the proton.. He believed that the total mass in atom is uniform in all atom's particles.
Here is when part 1 of john dalton's theory is disproven because it states atoms are indivisible when thomson learned there.. Dalton's atomic theory stated that the atom was indivisible and indestructible. Here is when part 1 of john dalton's theory is disproven because it states atoms are indivisible when thomson learned there. Dies ist im gegensatz zur heutigen auffassung, bei der die äußeren elektronen die chemischen eigenschaften des elements bestimmen. 10.06.2017 · since the atomic model constructed by jj thomson was one of the earliest atomic theory, it's understandable that his atomic theory still contains some flaws. 27.09.2021 · in 1900, jj thomson propounded the presence of a negatively charged particle inside an atom, equal in magnitude but opposite in sign to that of the proton. There were some inconsistency in his atomic theory and his atomic model. Curiosity arose as to how these particles are arranged within an.. Dies ist im gegensatz zur heutigen auffassung, bei der die äußeren elektronen die chemischen eigenschaften des elements bestimmen.

In 1897, thomson claimed the basic body of an atom is a sphere that contains electrons (tiny particles within an atom that create a negative charge) and a positively charged jelly around the electrons that neutralize the charge of the electrons... In magnitude the negative and the positive charges are equal. Here is when part 1 of john dalton's theory is disproven because it states atoms are indivisible when thomson learned there. He believed that the total mass in atom is uniform in all atom's particles. Die lichtemission der atome führte thomson auf schwingungen des elektronensystems zurück, die durch äußere störungen angeregt werden. 10.06.2017 · since the atomic model constructed by jj thomson was one of the earliest atomic theory, it's understandable that his atomic theory still contains some flaws. 27.09.2021 · in 1900, jj thomson propounded the presence of a negatively charged particle inside an atom, equal in magnitude but opposite in sign to that of the proton.

So, the whole atom is electrically neutral. 10.06.2017 · since the atomic model constructed by jj thomson was one of the earliest atomic theory, it's understandable that his atomic theory still contains some flaws. He believed that the total mass in atom is uniform in all atom's particles. There were some inconsistency in his atomic theory and his atomic model.. Here is when part 1 of john dalton's theory is disproven because it states atoms are indivisible when thomson learned there.
/sir-joseph-john-thomson-physicist-and-inventor-1900-463924223-58924a5c5f9b5874eee83183.jpg)
In 1897, thomson claimed the basic body of an atom is a sphere that contains electrons (tiny particles within an atom that create a negative charge) and a positively charged jelly around the electrons that neutralize the charge of the electrons.. An atom consists of a positively charged sphere, and it embeds the electrons. Here is when part 1 of john dalton's theory is disproven because it states atoms are indivisible when thomson learned there. Dies ist im gegensatz zur heutigen auffassung, bei der die äußeren elektronen die chemischen eigenschaften des elements bestimmen. Curiosity arose as to how these particles are arranged within an. He believed that the total mass in atom is uniform in all atom's particles. There were some inconsistency in his atomic theory and his atomic model. Limitations of jj thomson model of atom. In 1897, thomson claimed the basic body of an atom is a sphere that contains electrons (tiny particles within an atom that create a negative charge) and a positively charged jelly around the electrons that neutralize the charge of the electrons. Jj thomson proposed the neutrality in the atom. But the discovery of electrons and protons inside the atom led to the failure of this theory. Die lichtemission der atome führte thomson auf schwingungen des elektronensystems zurück, die durch äußere störungen angeregt werden.

Jj thomson proposed the neutrality in the atom. Jj thomson proposed the neutrality in the atom.

There were some inconsistency in his atomic theory and his atomic model. In magnitude the negative and the positive charges are equal. He believed that the total mass in atom is uniform in all atom's particles. But the discovery of electrons and protons inside the atom led to the failure of this theory. There were some inconsistency in his atomic theory and his atomic model.

In 1897, thomson claimed the basic body of an atom is a sphere that contains electrons (tiny particles within an atom that create a negative charge) and a positively charged jelly around the electrons that neutralize the charge of the electrons. Dies ist im gegensatz zur heutigen auffassung, bei der die äußeren elektronen die chemischen eigenschaften des elements bestimmen. So, the whole atom is electrically neutral. Die lichtemission der atome führte thomson auf schwingungen des elektronensystems zurück, die durch äußere störungen angeregt werden. Limitations of jj thomson model of atom. But the discovery of electrons and protons inside the atom led to the failure of this theory. Dalton's atomic theory stated that the atom was indivisible and indestructible. Curiosity arose as to how these particles are arranged within an. Here is when part 1 of john dalton's theory is disproven because it states atoms are indivisible when thomson learned there. Here is when part 1 of john dalton's theory is disproven because it states atoms are indivisible when thomson learned there.

In 1897, thomson claimed the basic body of an atom is a sphere that contains electrons (tiny particles within an atom that create a negative charge) and a positively charged jelly around the electrons that neutralize the charge of the electrons... 10.06.2017 · since the atomic model constructed by jj thomson was one of the earliest atomic theory, it's understandable that his atomic theory still contains some flaws. An atom consists of a positively charged sphere, and it embeds the electrons. So, the whole atom is electrically neutral. Dalton's atomic theory stated that the atom was indivisible and indestructible.. 27.09.2021 · in 1900, jj thomson propounded the presence of a negatively charged particle inside an atom, equal in magnitude but opposite in sign to that of the proton.

Die lichtemission der atome führte thomson auf schwingungen des elektronensystems zurück, die durch äußere störungen angeregt werden.. Here is when part 1 of john dalton's theory is disproven because it states atoms are indivisible when thomson learned there... Curiosity arose as to how these particles are arranged within an.